Session 4: Immune defences against infectious diseases
Use 'Print preview' to check the number of pages and printer settings.
Print functionality varies between browsers.
Printable page generated Monday, 27 April 2026, 7:42 PM
Session 4: Immune defences against infectious diseases
Introduction
In this part of the course you'll look at the body’s natural defences against pathogens – the infectious agents that cause disease. We describe how physical and chemical barriers keep pathogens from entering the body and how the immune system uses a range of specially adapted cells and molecules to attack pathogens that get past these barriers. One of these cell types is known as a B cell and it plays a vital role in producing antibodies to fight infection.
Watch the following video to learn more about this process and the role of B cells in producing an immune response to vaccination.
Transcript
CLAIRE ROSTRON: Antibodies are vital in our fight against infectious diseases because they're created by the body to recognise the presence of a pathogen invader. They're created by specialised parts of our immune system, called B cells. So called because they're found in the bone marrow.
Antibodies are very large molecules. While there are several different types, the most common of these found in the human body are made up of around 25,000 atoms. This large array of atoms tends to bunch and fold itself up to form crevices and bumps.
Every type of pathogen has at least one-- but often many more-- unique molecules known as antigens in their structure. Antigens are the parts of the pathogen that can be detected by antibodies, which work a bit like a pattern recognition system. We can imagine an antibody to be a bit like a lock devised from the crevices and bumps, such that only the right--shaped key will comfortably fit into the lock. But in this case, only the right antigen will fits into the bumpy and creviced antibodies that our bodies make.
The B cells, like typical animal cells, have cell membranes and they carry antibody molecules embedded by their tails in the cell membrane, with the antigen-binding sites facing outwards into the bodily environment. An antibody typically has two binding sites. When an antigen binds to the antibody at either site, it triggers the B cell to produce many more at the same antibody, which are then released into the bloodstream to circulate the body and detect further pathogens.
Antibodies themselves cannot kill pathogens. So strictly speaking, they cannot fight infection. They're simply a signalling mechanism that says to other cells of the immune system, here is a pathogen. Come and get it.
But their signalling is vital for immunity, since once the B cells have developed antibodies specific to an antigen, they can do so again but much faster. And this means that the recognition of a pathogen invader is much more efficient and the body cells can kill this pathogen invader before it replicates extensively and takes hold.
In this Open Learn course, you'll learn more about the B cells and other cells of the immune system.
[MUSIC PLAYING]
4.1 Natural barriers against pathogens
The physical and chemical barriers that prevent pathogens from getting into our body tissues in the first place are often overlooked. As Figure 1 shows, the most comprehensive barrier is the waterproof layer of skin that covers the body’s surface. Human skin keeps most pathogens out as long as it remains intact. The speed with which a cut or graze can become infected is a reminder of the protection we normally get from our skin.
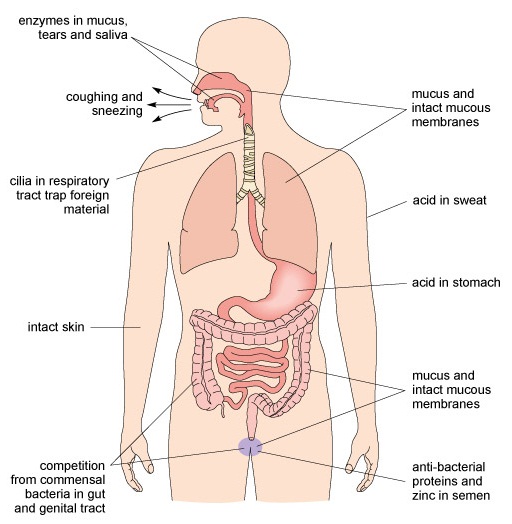
This is a diagram of the human body annotated with descriptions of the various barriers against infection. These comprise: intact skin and the acid in sweat; coughing and sneezing which expel infectious agents; enzymes in mucus, tears and saliva, in the nose and mouth; the mucus and intact mucous membranes of the respiratory tract; cilia in the respiratory tract, which trap foreign material; acid in the stomach; the mucus and intact mucous membranes of both the gut and the genital tract; competition from commensal bacteria in the gut and genital tract; and, in males, antibacterial proteins in semen.
Vector-borne infections (e.g. malaria) are transmitted by biting invertebrates (e.g. mosquitoes) penetrating the skin when taking a blood meal. Humans are also vulnerable to invasion by pathogens in the air, food, water and soil, or during physical contact with infected people, some animals (e.g. pigs, dogs, poultry) or their faeces. As Figure 1 illustrates, the inner surface of the respiratory system (nose, throat, airways and lungs), stomach, intestines, bladder and reproductive tract are lined with membranes that secrete jelly-like mucus, presenting a barrier against pathogens entering our tissues via these routes. Microscopic hairs called cilia [sill-ee-ah] line the respiratory system and ‘beat’ in unison to shunt mucus containing trapped pathogens towards the nose and mouth, where they can be expelled by coughing and sneezing, or swallowed into the stomach where acid destroys them.
Figure 1 refers to competition from commensal bacteria (sometimes referred to as ‘friendly bacteria’) in the gut and reproductive system. Commensal bacteria are non-pathogenic inhabitants of the gut and the reproductive system. They occupy space that could otherwise be colonised by pathogenic species, and they use nutrients for their own growth, thereby reducing the resources available for pathogens to multiply and cause disease.
Chemical barriers against infection include enzymes in tears, saliva and mucus that break down the surface of bacteria. The acid in sweat and in the stomach kills cellular pathogens and there are anti-bacterial proteins in semen (the fluid that contains male sperm).
The more complex mechanisms of the immune system are only needed if pathogens breach these physical and chemical barriers.
4.2 The immune response to infection
The human immune system is an extremely complex network of interacting cells and biological molecules. Our aim here is simply to give you an overview of how an immune response to infection develops, without going into too much detail. It occurs in three overlapping stages, the first of which is triggered when body cells are damaged.
When tissues are injured, the damaged cells release chemicals that trigger the sequence of events described as inflammation. It occurs in response to any type of injury, such as a blow or a cut, an insect bite, or damage caused by pathogens multiplying in body tissues. Inflammation has four characteristic effects at the site of an injury, the first two of which are visible around the splinter shown in Figure 2:
- swelling
- redness
- heat
- pain.
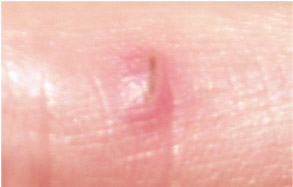
The inflamed area shows these signs because the local blood vessels dilate (get wider), increasing blood flow into the injury site, so it looks red as well as feeling warmer than the surrounding tissue. The walls of the blood vessels near the injury become leaky, allowing fluid, defensive proteins and immune system cells (described shortly) to flood into the area, which becomes swollen as a result. One of the proteins released during the inflammatory response also makes the area more sensitive to painful stimuli, so inflamed tissue is sore to touch.
Sites of tissue injury are vulnerable to invasion by pathogens so the benefits of inflammation generally outweigh the discomfort it causes. Flooding the area with fluid dilutes any pathogens that are already present, and the local concentration of immune system cells and defensive proteins enables an immune response to begin more quickly.
4.3 Leukocytes: the cells of the immune system
Once the barriers to infection have been breached and inflammation has begun, the active agents of the immune system, the leukocytes [loo-koh-sites], get to work. Leukocytes are often described as ‘white blood cells’ to distinguish them from the red blood cells that transport oxygen around the body; however, calling them ‘blood’ cells is misleading because leukocytes roam throughout the body tissues and only spend part of their lives in the bloodstream. In fact, they spend more time in the lymphatic system (Figure 3), the network of fine tubules that collect tissue fluid from all over the body and return it to the bloodstream.
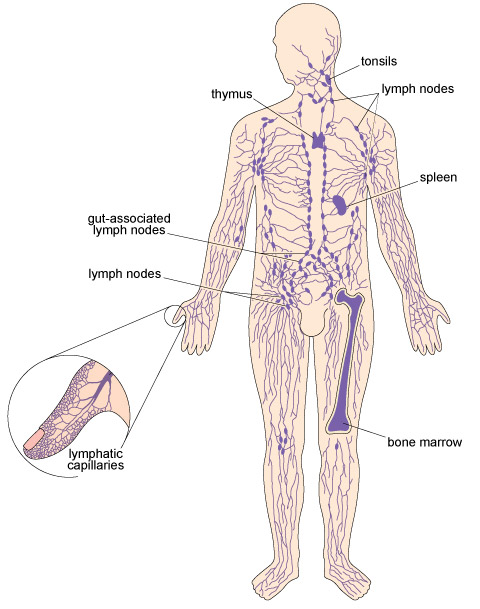
This figure is a simplified diagram of the human lymphatic system. It comprises a network of vessels which terminate in beds of tiny lymphatic capillaries; these are shown in detail in a blow-up diagram of the lymphatic system within the thumb. At locations along the lymph vessels are small oval-shaped structures called lymph nodes. These are distributed widely throughout the body and are clustered in the groin, under the arm and in the abdomen, in association with the gut. Other lymphoid structures present and their locations are the tonsils (at the back of the soft palate), the thymus (in the chest) and the spleen (in the upper left of the abdomen). There is also lymphoid tissue in the soft interior of the bones, called the bone marrow.
The lymphatic system includes specialised organs and tissues where leukocytes develop. During an immune response to pathogens, we may become aware of swollen lymph nodes (popularly called ‘glands’) in the neck, armpits or groin, which enlarge when the leukocytes they contain are multiplying near a site of infection.
Leukocytes can distinguish between ‘self’, the cells and proteins generated by the organism whose body they patrol, and ‘non-self’ (or ‘foreign’) material such as pathogens that originated outside the host’s body. Leukocytes are self-tolerant, i.e. they do not normally attack the host’s own cells or body proteins, but direct their actions only against non-self material that may pose a threat.
Although we have referred to ‘the’ immune response, as if it was just one thing, in fact, there are two types of immune response, distinguished as innate and adaptive immunity.
4.4 Innate immunity
All animals, even those with much simpler bodies than our own (e.g. parasitic worms) respond to tissue damage in ways that resemble inflammation in humans. They have cells similar to leukocytes and defensive proteins that flood into areas of tissue damage or infection. These leukocytes and proteins can defend the organism from pathogens because they recognise common patterns of molecules that occur in the structures of many different types of pathogens. These pathogen ‘signature’ molecules are known as PAMPs, or pathogen-associated molecular patterns.
The fact that PAMPs are commonly found in unrelated pathogens means that the leukocytes that recognise them cannot tell one type of pathogen from another. This non-specific immune response against pathogens is so widespread among animals that it is described as innate immunity (‘innate’ means ‘inborn’). Some texts use the alternative term ‘natural’ immunity.
The leukocytes involved in innate immunity are of two general types, each with a different action against pathogens:
- Cytotoxic [sigh-toh-tox-ik] leukocytes, which simply means ‘cell poisoning’ (the prefix ‘cyto’ denotes a cell). These leukocytes have various methods of attaching to the outside of a pathogen and releasing destructive chemicals onto its surface. Worm larvae, bacteria and protists can all be killed this way.
- Phagocytic [fag-oh-sit-ik] leukocytes (the prefix ‘phago’ comes from a Greek word meaning ‘to eat’), often abbreviated to phagocytes [fag-oh-sigh-tz]. These leukocytes engulf small pathogens such as bacteria, drawing them into the cytosol where destructive chemicals break them down. This action is termed phagocytosis [fag-oh-sigh-toh-siss].
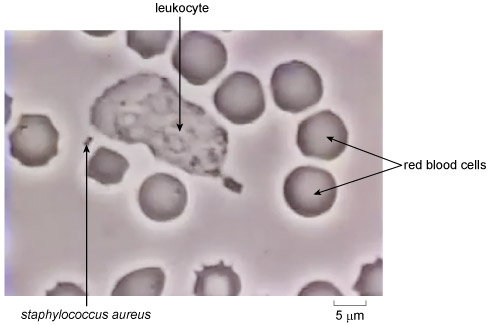
Figure 4 identifies a phagocyte, red blood cells and two tiny spherical bacteria (Staphylococcus aureus) joined together – this species is often found in ‘strings’ of two or more linked cells. Red blood cells are fragile and some have begun to disintegrate in these culture conditions. Note that red blood cells are also known as erythrocytes [air-rith-roh-sigh-tz].
Viruses are small enough for a leukocyte to engulf if the viruses are floating ‘free’ in the bloodstream or tissue fluids, but once in the body, viruses rapidly become hidden from phagocytic leukocytes because they must infect the host’s own cells in order to make new virus particles. Can you suggest how this protects the virus from the innate immune response?
Once inside the host’s cells, they are hidden from the self-tolerant phagocytic leukocytes, which can only recognise and engulf material that originated outside the host’s body. (As you will see later, adaptive immunity has a solution to this problem.)
The anti-pathogen activities of certain specialised proteins are important contributors to the innate immune response. They include proteins that accelerate inflammation, target leukocytes onto pathogens or make host cells resistant to invasion by viruses. Their concentration increases rapidly in the bloodstream during an infection and this rise can be detected in blood tests as a diagnostic sign of infection.
In summary, all animals have innate immunity based on cells similar to human leukocytes and defensive proteins that defend the organism against pathogens ‘in general’ in a non-specific way, i.e. these defences cannot distinguish between one type of pathogen and another. Humans and other warm-blooded animals have an additional defensive capability called adaptive immunity, which differentiates specifically between pathogens, as the next section describes.
4.5 Adaptive immunity
Adaptive immunity is due to the actions of two types of specialised leukocytes, known as T cells and B cells. (If you are interested, the letters denote ‘thymus’ and ‘bone marrow’, the tissues where each of these leukocytes mature.) We will describe their individual contributions to the adaptive immune response shortly, but first we focus on the most striking difference between innate and adaptive immunity. The clue lies in the word ‘adaptive’.
T cells and B cells have recognition methods that distinguish between different pathogens (e.g. different species of bacteria), and they adapt during their first encounter with a particular pathogen. The second time they meet it in the body, the adaptive response begins earlier, lasts longer and is more effective than it was on the first occasion. You can learn more about this by watching the following animation. (If you do not wish to see closed captions, use the 'CC' (captions) button to remove or reveal the subtitles.)
Transcript
INSTRUCTOR: The immune system is said to be adaptive because, after the first encounter with a pathogen, it can develop a much faster response to repeat infection with the same pathogen. This adaptive response is important for vaccination and immunisation. Let's take a closer look at some graphs that illustrate this phenomenon.
This graph shows the first encounter with a pathogen, which might be, for example, the chicken-pox virus. If we chart the number of antibodies and leukocytes the body produces on the vertical axis over time on the horizontal axis, we can see that, after infection at time 0, it takes 10 days for antibody and leukocyte numbers to start increasing. This increase in production of antibodies and leukocytes lasts for just over 15 days.
Now let's take a look at the secondary adaptive immune response by plotting this on the same chart. This occurs with a repeat infection by the same pathogen. In our example, this would be a repeat contraction of the chicken-pox virus. In this case, after infection at time 0 it takes less than 5 days for antibody and leukocyte numbers to start increasing.
The production of antibodies and leukocytes lasts for over 30 days. And it is a noticeably larger increase, compared to the increase seen in our primary response. How is this possible?
The cells of our immune system that are responsible for this phenomenon are known as "B cells." B cells are designed to recognise only a specific pathogen, and so we have billions of them in our bodies. During a primary encounter with a pathogen, the B cell binds to the pathogen via receptors and eventually becomes activated. At this point, it starts dividing, producing copies of itself.
Some of these new cells become "plasma cells." This is the name given to the cells that function as antibody factories, producing antibodies that recognise the pathogen and flag it for destruction by other cells of the immune system. However, these plasma cells only live for a few days.
In contrast, some of the clone cells become memory cells, with a life span of decades. They circulate in the bloodstream, ready to produce antibodies much more quickly when they next encounter the same pathogen. It's the memory cells that produce the secondary immune response.
Thanks to scientific experimentation, we now know that it's possible to deliberately administer a pathogen to generate an immunological memory by the production of memory B cells. It's this process that underlies vaccination and other forms of immunisation.
So, there is a much faster and increased response to a subsequent encounter with a pathogen and this demonstrates the adaptability of the immune system. This response is due to the production of long-lived memory cells that circulate in the body after the primary adaptive immune response subsides. These memory cells are specifically programmed to recognise the same pathogens that triggered the primary response if they ever get into the body again. You will learn much more about these later in this session.
Overall then it is to be expected that one of the appropriate immune system responses to infection is an increase the concentration of leukocytes in the blood circulation. This expected response can actually be tested in a laboratory by taking a blood sample from an individual who is suspected to be suffering from an infection. Blood from the sample is then smeared onto a microscope slide and air dried, and the sample can then be viewed at different magnifications using a light microscope to enable the number of leukocytes to be counted. In the activity in the next section you can test for the presence of the suspected infection by counting leukocytes in blood samples using our digital microscope.
4.6 Counting leukocytes in blood samples
The Digital Microscope Leukocyte Counting Activity is one of a number of interactive, practical science resources that you can access from the OpenScience Laboratory website: a collaborative initiative of The Open University and The Wolfson Foundation. Here you will find investigations, tools and activities coving a broad range of scientific fields, including health science, astronomy and earth science many of which are also built into the learning experience on our modules.
When you first access the tool you will be prompted to either sign in (if you are already an Open University student), or to register with your email address to create an account free of charge. This only takes a few minutes to do and the website will guide you through the process.
Note that the interactive activities on the OpenScience Laboratory website require a modern web browser, such as Mozilla Firefox, Google Chrome, Apple Safari or Microsoft Internet Explorer 9 or later.
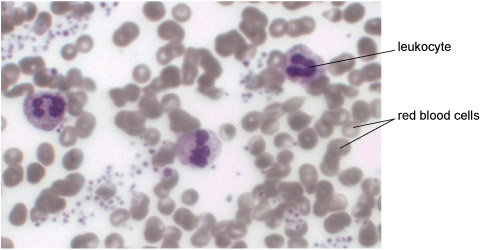
The easiest way to look at leukocytes is to take a tiny quantity of blood and smear it onto a glass slide, air dry the cells and then treat them with a dye that stains leukocytes blue/purple. Figure 5 shows several leukocytes that have been prepared in this way, with some red blood cells around them, which appear grey. Not all leukocytes look exactly like the ones in Figure 5, but the most numerous type in the bloodstream – the neutrophils [nyoo-troh-fillz] – have this general appearance. The dark blue/purple shapes inside each neutrophil are the connected lobes of its irregularly shaped nucleus.
4.6.1 The digital microscope
In the following activity, you will use the digital microscope to count leukocytes in microscope slides of blood smears that look like Figure 6. This figure shows a magnified blood sample from a healthy person (termed the ‘Normal’ sample). A second slide (not shown here) has blood from someone whose immune system was believed to be fighting a severe infection when the sample was taken. We have labelled this one ‘leukocytosis’ [loo-koh-sigh-toh-siss]. Leukocytosis refers to an increase in the total number of leukocytes of all types circulating in the bloodstream.
When you have counted the leukocytes in your two blood samples, you can compare your counts and decide whether there is evidence to suggest that the sample labelled ‘leukocytosis’ was taken from an individual who was indeed suffering from an infection.
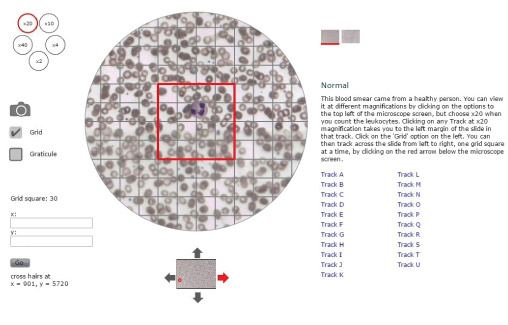
Note the following features in Figure 6, which will help you to navigate the digital microscope when you access it online:
- You can switch between the ‘Normal’ and the ‘Leukocytosis’ slides by clicking on the squares to the top right of the microscope screen.
- The small circles to the top left of the microscope screen show the magnification factors you can choose for viewing the blood smears. They range from ×2 (twice the actual size of the cells) to ×40. Click on the circle labelled ×20 to choose the optimum magnification for this activity.
- When you click on the small square labelled ‘Grid’ on the left of the microscope screen, a matrix of faint grey lines is superimposed on the blood smear with a large red square in the centre of the grid. Your task is to count any leukocytes in the red square as you move it across the blood smear.
- The slides have been divided into horizontal sections labelled Track A to Track U, which are listed on the right of the microscope screen. It is not necessary to count all tracks on a slide although you can do if you wish. We suggest you just choose one track as a representative sample of the whole slide. When you click on a track (e.g. Track A) with the magnification set at ×20, the microscope screen shifts automatically to the left margin of the slide in that track.
- Notice the up/down/left/right arrows around the small image of the slide below the main microscope screen. If you click on the red arrow, it moves the slide to the right by one red grid square. This enables you to move across the blood smear one red grid square at a time until you reach the other side of the slide.
4.6.2 Testing for the presence of infection
You'll now use the digital microscope to test for the presence of infection.
Activity 1 Using the digital microscope
Before you access the digital microscope read through the following instructions for the leukocyte counting activity carefully:
- Remember to increase the magnification to help you count leukocytes by clicking on the circle labelled ‘×20’ in the top left corner of the screen.
- Locate the square button labelled ‘Grid’ below the camera symbol on the left of the screen and click on it so that the grey counting grid appears to help you count systematically.
- Now choose which track you wish to count (A-U) and click on the appropriate letter. This will take your viewer (a red square) to the far left of the correct track so that you can begin counting.
- Count the number of leukocytes (if any) in the red grid square on the far left of your track noting down the number of leukocytes into the grid square number 1 of Table 1 (see below).
- Return to the digital microscope and click once on the red arrow pointing to the right in the small box below the blood smear. This moves the blood smear an exact distance to the right, so the red square is now around Grid square 2 in your track.
- Count the leukocytes (if any) in Grid square 2 and enter the number into Grid square number 2 in Table 1.
- There are 29 complete red grid squares in every track, plus ‘half’ a grid square (numbered 30) at the right-hand edge of the track, which you should include in your counts. You can then keep moving the slide one red grid square to the right by clicking once on the red arrow and counting and recording the number of leukocytes in each red square until you finally reach the right-hand margin of the slide.
- Finally, add up the total number of leukocytes in your track of the Normal blood smear and enter the total in Table 1. Once you have completed a table for your one of the slides (e.g. the normal slide), repeat the process for the other slide (e.g. the leukocytosis slide).
Table 1 Table for recording the number of leukocytes in your track of the Normal blood smear.
| Normal blood smear track (letter) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grid square number | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 |
| Number of leukocytes | |||||||||||||||
| Grid square number | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | 25 | 26 | 27 | 28 | 29 | 30 |
| Number of leukocytes | |||||||||||||||
| Normal Track total: | |||||||||||||||
Note that some leukocytes will inevitably sit across the margin of a grid square, as in the example in Figure 7.

This is a very common occurrence in biological investigations where large numbers of ‘subjects’ (e.g. cells or organisms) are counted systematically using a grid to ensure that every cell or organism is accurately recorded.
If a leukocyte is ‘divided’ by a grid line on the left or the right margin of the red square, it is still in ‘your’ track so it will count towards your track total no matter which grid square you decide to record it in Table 1. But if a leukocyte is partly inside and partly outside the upper or lower margin of a red square (as in Figure 3), you must make a decision about whether to count it in ‘your’ track or exclude it as belonging to the track above or below yours.
Now access the digital microscope leukocyte counting activity on the Open Science Laboratory website. (If you don't already have one, you'll need to sign up for a free Open University account in order to access this. The website will guide you through the process.)
Once you have counted the number of leukocytes for each slide, compare the two values. Do they differ? If so which one is larger? Does this support the suspected infection in the participant whose sample was taken for the ‘leukocytosis’ slide?
Discussion
You should have found that the leukocytosis slide contained more leukocytes than the normal slide. This supports the suspected infection in the participant whose sample was taken for the leukocytosis slide. Note that if you opted to count more than one track on each slide you would also have reached a more reliable answer to this question about suspected infection. This is because the larger the area you count the more representative this is of the number of leukocytes that are present in the blood as a whole.
You might also wish to reflect on how straightforward you found this counting process. Did you find it easy to decide whether to count a borderline cell or not? Was it easy to spot a leukocyte using the purple staining colour or was there an element of subjectivity involved?
We have designed this microscope activity to make it as close to the reality of cell counting on a microscope as possible. As such the subjectivity you may have applied to the counting process would be representative of the real experience of cell counting. Whatever subjective criteria you applied when deciding whether to count a leukocyte or not should have been applied consistently.
4.7 Chickenpox: adaptive immunity in action
Chickenpox infection demonstrates how effective the adaptive immune response can be in preventing reinfection with the chickenpox virus. Children who have suffered from chickenpox (Figure 8) and recovered are unlikely ever to develop it again, because they have circulating memory cells specifically programmed to recognise chickenpox viruses. These memory cells are unable to recognise any other pathogens, but they react swiftly and effectively if the chickenpox virus gets into the body again. These memory cells direct the more vigorous secondary adaptive immune response, which produces many new T cells and B cells programmed specifically to attack chickenpox viruses.
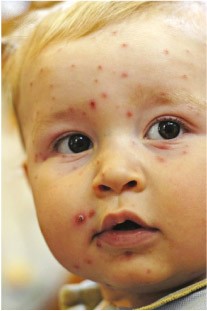
The secondary adaptive immune response is usually so effective that the person doesn’t become ill and may never know that they have been infected by chickenpox viruses for a second time. When the secondary response subsides, even more memory cells that recognise chickenpox viruses as their specific target are left in circulation, providing lifelong protection against this pathogen to almost everyone who suffered this disease as a child.
Chickenpox usually resolves without treatment and because it is extremely rare for someone to develop it a second time, many countries (including the UK) do not routinely vaccinate children against this virus unless they have a deficient immune system. But many other pathogens produce far more serious diseases during their first encounter because the infection develops faster than the primary adaptive immune response can react against it. In a later section, we explain how vaccination can enhance the secondary adaptive immune response and prevent an infection from developing.
But first we have to explain how leukocytes in the adaptive immune system recognise each type of pathogen so specifically.
4.8 Antigens and the specific recognition of pathogens
Every type of pathogen has at least one (often many more) unique molecules known as antigens in their structure. In addition to the PAMPs (pathogen-associated molecular patterns) shared by many different pathogens, each type of pathogen also has its own unique distinctive antigens. Each individual T cell and B cell (the leukocytes responsible for adaptive immunity) is programmed to recognise just one specific antigen, so it follows that each T or B cell can usually recognise only one type of pathogen, or at most two or three closely related pathogens that have very similar antigens. Recognition of an antigen by these adaptive leukocytes triggers an immune response against only those pathogens with that antigen in their structure. The political slogan ‘One person, one vote’ springs to mind as an analogy for ‘One adaptive leukocyte, one target’!
We haven’t yet answered the question of how T cells and B cells recognise antigens. Each of these leukocytes carries receptor molecules (often abbreviated to ‘receptors’) on its outer cell membrane. Receptors are very large molecules containing hundreds or even thousands of atoms. As a consequence, they fold up into very complex 3D shapes with many troughs, crevices, humps and hollows, creating a molecular landscape that is unique for each receptor molecule.
A particular type of receptor molecule on the surface of a T cell or B cell can only recognise an antigen that has a 3D shape which is the ‘mirror-image’ of this receptor, so the two molecules can fit together like a key in a lock. In fact, the contact area between the receptor and the antigen involves only a tiny part of each molecule (Figure 9), but this is enough to hold them together long enough to trigger changes in the leukocyte.
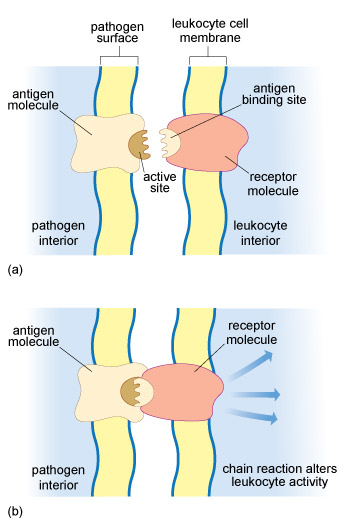
This is a schematic diagram showing part of a leukocyte cell membrane with a receptor molecule in its surface. A small region of the outward-facing part of the receptor is identified as the binding site. Close to the leukocyte cell membrane is a pathogen surface in which there is an antigen molecule. A small part of the antigen, the active site, has a shape that fits into the binding site of the receptor. Part (b) shows the receptor bound to the antigen, and this binding initiates a chain reaction that alters the leukocyte’s activity.
Each T cell and B cell carries many identical copies of a single antigen receptor, so an individual T or B cell can only bind to pathogens that display the corresponding antigen. For example, a T or B cell with receptor molecules that fit an antigen found only in the structure of malaria protists is unable to recognise any other pathogen as a target for an adaptive immune response – it cannot bind to the antigens of (for example) TB bacteria or polio viruses.
At least 10 million different antigen receptors, each with a unique 3D shape, are necessary to recognise all the pathogens an individual may encounter in a lifetime. How this vast array of antigen receptors is generated by T and B cells, each of which carry just one antigen receptor shape, is beyond the scope of this course, but our survival depends on this marvellous phenomenon.
By contrast, the leukocytes responsible for innate immunity have receptor molecules that recognise PAMPs, which many different pathogens have in common. This is why innate leukocytes have a non-specific response to infection – in a sense, all pathogens ‘look alike’ to them, so they are incapable of forming a ‘memory’ of having encountered a pathogen before.
We said earlier that we would explain the different actions of T cells and B cells in the adaptive immune response to infection. We start by considering B cells in the next section, which produce proteins called antibodies.
4.9 Antibodies and B cells
Antibodies are very large proteins that contribute to adaptive immunity. There are several types, but the most abundant antibody molecules in humans each contain about 25 000 atoms. A distinguishing feature of antibodies is that their structure includes at least two binding sites for antigens. Most antibodies resemble the simplified representation in Figure 10.
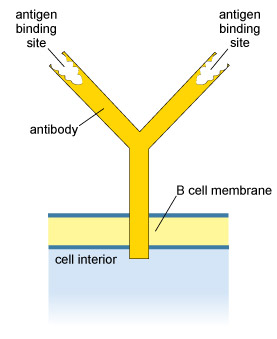
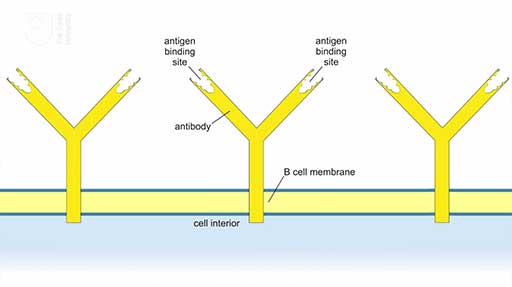
This is a schematic diagram showing the Y-shaped antibody molecule with its ‘stem’ embedded in the B cell membrane and extending a small distance into the cell interior. In each of the two ‘arms’ of the molecule is a binding site for its specific antigen
B cells produce antibodies and also use them as their antigen receptors. The B cells carry antibodies embedded by the ‘tail’ in their outer cell membrane, with the binding sites facing outwards (Figure 10). This enables the B cell to bind to antigens that fit the binding sites in the antibodies it carries on its surface. This binding event is essential (but not sufficient on its own) to activate B cells into making a lot more antibody molecules that recognise the same antigen. These antibodies are released by the B cells and circulate in the bloodstream, tissue fluids and the lymphatic system. Antibodies are also abundant in the mucus membranes lining the respiratory system, the gut and the reproductive system, i.e. the sites in the body in contact with substances such as air, food, drinking water and sexual fluids that could contain pathogens.
Antibodies are often portrayed in the media as if they were ‘magic bullets’ that attack pathogens, but in fact they are more like ‘waving flags’ with a message that reads ‘here is a pathogen – come and destroy it’. When antibodies bind to a pathogen, they simply label it for destruction by leukocytes with the innate ability to phagocytose (engulf) it, or cytotoxic (cell-killing) leukocytes and defensive proteins. You can think of them as recruiting the cells and defensive proteins of the innate immune system to join the attack.
We conclude this tour of adaptive immunity by describing the T cells.
4.10 T cells in adaptive immunity
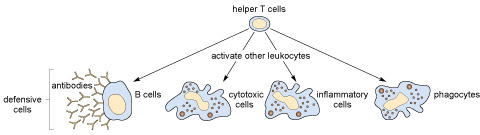
There are two types of T cells with different roles in adaptive immunity. The cytotoxic T cells release destructive chemicals onto their target’s outer surface in much the same way as the cytotoxic leukocytes do in an innate immune response. But there is one crucial difference. Cytotoxic T cells are programmed to kill the body’s own cells that have become infected by viruses or by the few types of bacteria and single-celled pathogens that can ‘hide’ inside the cells of their host (Mycobacterium tuberculosis, the bacteria that cause TB, can do this). Without the cytotoxic T cells, we would be particularly susceptible to infectious diseases caused by these pathogens.
The other T cell type is called the
This is a schematic diagram showing the Y-shaped antibody molecule with its ‘stem’ embedded in the B cell membrane and extending a small distance into the cell interior. In each of the two ‘arms’ of the molecule is a binding site for its specific antigen
If you have seen documentaries or read reports about HIV/AIDS, you have possibly heard that HIV (the human immunodeficiency virus) infects and ultimately destroys the helper T cells.
Why does the destruction of helper T cells by HIV leave a person susceptible to many other infections?
When the number of helper T cells declines to a low level, they cannot activate the other leukocytes to act effectively against other pathogens, so the person becomes susceptible to infections they could otherwise have overcome. Eventually the person develops AIDS – acquired immune deficiency syndrome – because their immune responses have become so deficient.
Next we explain how our defences against pathogens can be enhanced by vaccination, and consider some of the problems that limit the ability of vaccines to protect us from infection.
4.11 Vaccination
Vaccination relies on the development of immunological memory for its protective effect. Vaccines contain killed pathogens, or extracts from pathogens, or modified strains of pathogens that are no longer harmful. For example, the MMR vaccine contains weakened (the technical term is ‘attenuated’) variants of the three viruses that cause measles, mumps and rubella (Figure 12).
Why do you think most vaccines are given in two (or more) doses spaced at intervals of weeks or months?
The first dose triggers a primary adaptive immune response, but this doesn’t reach a high level and subsides over time. The second dose triggers a much more vigorous secondary response, generating a lot of memory cells, which remain on patrol to protect the child if he or she is subsequently infected with live pathogens of the same type as used in the vaccine.
Note that each of the memory cells and the T cells and B cells generated in an adaptive immune response are specifically directed against just one type of pathogen or a very closely related strain. Vaccination with MMR vaccine is only protective against measles, mumps and rubella infection.
Despite decades of effort, medical science has so far been unable to produce effective vaccines against a number of important infectious diseases, including HIV/AIDS and other sexually transmitted infections (although trials of several candidate vaccines are underway). Vaccines against malaria have shown some potential, but they only protected about half the vaccinated children in large-scale African trials (RTS,S Clinical Trials Partnership, 2012). There are no effective vaccines against infections caused by any other single-celled or multicellular parasites.
Some vaccines are effective only for a relatively short time, e.g. cholera vaccines give protection for only around six months. Others, such as the tetanus vaccine, need repeated booster doses. Annual influenza vaccinations are offered to vulnerable groups in the population because influenza viruses alter their antigens over time.
Why can’t vaccines prepared from last year’s influenza viruses be guaranteed to give protection against currently circulating flu strains?
Vaccines prepared from earlier strains of influenza viruses will not contain any of the altered antigens that currently circulating strains may have developed; last year’s vaccine cannot boost adaptive immunity against viruses with these new antigens.
The second practical challenge is organising and delivering mass vaccination programmes in low- and middle-income countries, especially in remote areas and urban shanty settlements. The terrain may be difficult, transport and communication links are often poor, and communities may not be aware when vaccination teams arrive. Most vaccines have to be kept cold or they lose their potency, but maintaining vaccines at 2–8 °C while vaccinating thousands of children in warm climates without adequate refrigeration requires ingenuity (Figure 13).

Thirdly, education programmes struggle to overcome basic lack of knowledge about vaccination, especially in communities where literacy rates are low and poverty is a daily fact of life. A survey of over 27 000 children in 24 African countries concluded that poor parental education and lack of access to information were critical factors in areas of low vaccine coverage. Only 71% of these children had received the full series of three vaccinations against diphtheria, tetanus and pertussis (whooping cough) by their first birthday, leaving an estimated six million African children unprotected (Wiysonge et al., 2012).
4.12 Opposition to vaccination
Another major challenge is parental opposition to vaccination because of concerns about vaccine safety. Some parents are understandably anxious about taking a healthy child to be given a vaccine derived from infectious agents. Most vaccines are injected, which can be a frightening and painful experience for a child. Of even greater concern is that some children experience a negative reaction to a vaccine, most often inflammation around the injection site and/or a mild fever lasting a day or two, but rare instances of more severe reactions such as convulsions sometimes occur.
Hesitancy about vaccinating a child is increased if parents have no experience of the disease the vaccine prevents, because it has become rare in communities with high vaccine coverage. Some parents conclude that vaccination is unnecessary and might expose their children to an avoidable risk.
Negative rumours about a vaccine are another deterrent. For example, the myth that vaccination is a Western plot to sterilise Muslim children has deterred parents in northern Nigeria, Pakistan and Afghanistan from allowing their children to be vaccinated against polio (Figure 14). Terrorist attacks have occurred against polio vaccination teams in all three countries – the only remaining locations where polio had not been eradicated by the WHO target of 2015.
An inevitable consequence of falling vaccination rates in a community is an increase in infections, sometimes with fatal outcomes. In 2014, the WHO warned that progress on eliminating measles had stalled and the number of deaths had begun to rise – from 122 000 in 2012 to 145 700 in 2013 (WHO News release, 2014). Dr Peter Strebel, from the WHO Department of Immunization, Vaccines and Biologicals warned that:
Countries urgently need to prioritize maintaining and improving immunization coverage. Failure to reverse this alarming trend could jeopardize the momentum generated by a decade of achievements in reducing measles mortality.
Even in countries where over 90% of children are fully vaccinated, uptake rates can plummet under pressure from negative rumours. For example, false claims in 1998 that the MMR vaccine caused autism led to a sustained fall in vaccinations in the UK, followed by a significant rise in measles cases and, to a lesser extent, mumps. The largest outbreak of measles around Swansea in Wales in 2012–13 caused more than 1200 cases and one death (Public Health Wales, 2013). Once parents saw the reality of measles infection and the misery and discomfort it causes, a rapid increase in uptake of the MMR vaccine followed the outbreak. The memory of infection fades in well-protected communities, until an outbreak reminds everyone of how devastating infectious diseases can be.
Session 4 quiz
This quiz allows you to test and apply your knowledge of the material in Session 3.
Open the quiz in a new window or tab then come back here when you're done.
Summary to Session 4
Having completed this final session of the course you should now be familiar with the basic features of a human being’s defences against pathogens. The first lines of defence are the physical barriers formed by the skin and secreted mucus, and the initial reaction to a breach of these defences is inflammation. We have introduced you to leukocytes, the key family of cells involved in the two types of immunity: innate and adaptive.
- Innate immunity involves the targeting of non-specific structures, known as PAMPS, on the surface of pathogens, and involves cytotoxic and phagocytic leukocytes.
- Adaptive immunity involves B cells and cytotoxic T cells, two types of leukocyte with receptors that bind to specific molecules on the surface of individual types of pathogen, as well as helper T cells that release signals to boost the immune response.
You have also learned that adaptive immune responses are boosted with repeated exposure to the pathogen due to the remarkable phenomenon of immunological memory. Importantly, immunological memory can be used by health care professionals to prevent disease through the process of vaccination. By exposing a person to an inactive form of a pathogen, that individual acquires an immunological memory of the pathogen. This means that if in future they become infected with the live pathogen they will launch a strong adaptive immune response and the pathogen is quickly destroyed.
As we have outlined, mass vaccination programs represent a key strategy in global health initiatives aimed at eradicating, or at least controlling, infectious diseases. Mass vaccination can be highly effective but, as you will now be aware, vaccine delivery is not straightforward; health workers often have to overcome ignorance and fear, as well as the logistical problems associated with so large a task.
Conclusion
In this free course on Infection and immunity you have learned that from prion to protists, via all manner of pathogenic bacteria, viruses and parasites, it is clear that pathogens pose a major threat to human health. Through our immune systems we possess some effective weapons to fight off them off, but these aren’t fool proof; infectious diseases remain a major health care challenge.
However, with the careful use of the scientific method we can understand more about the nature of pathogens and use this information to develop new strategies to prevent – and perhaps eradicate – the deadly threat they pose.
- Infectious diseases kill around 10 million people every year worldwide and are caused by pathogens.
- There are more than 1400 different pathogens that cause human diseases, but only about 20 cause two-thirds of human deaths from infection.
- There are many types of pathogen, including multicellular parasites, single-celled protists, yeasts and bacteria, as well as viruses and prions.
- Pathogens can be transmitted directly or indirectly. Direct methods included transmission by touch, sexual transmission and mother-to-child transmission. Indirect methods involve transmission via air, water or food, such as the faecal–oral route and environmental contamination.
- The underlying causes of infectious diseases include biological susceptibility, the social and economic conditions in which people live, and behaviours that influence the spread of infection.
- The symptoms of a disease can only be experienced by the person who suffers from them, whereas the signs of a disease can be observed by others.
- Infectious diseases may be acute, resulting in recovery or death within a few weeks, or chronic and slowly progressing over months or years; a chronic condition may include an acute episode.
- The application of the scientific method has been central in humankind’s fight against infectious disease, and is characterised by systematic observation, measurement, experiment, and the formulation, testing and modification of hypotheses.
- Early examples of the use of the scientific method include John Snow’s experiment of removing the handle from the Broad Street pump, which demonstrated that cholera was transmitted in contaminated water almost 30 years before bacteria were identified, and Edward Jenner’s smallpox inoculation experiment.
- A person’s defences against pathogens begin with physical and chemical barriers such as intact skin; when these are breached we rely on our immune system to fight infection.
- The immune system has two distinct branches – innate and adaptive immunity – each of which uses different types of leukocytes.
- The innate immune system is non-specific; the leukocytes involved cannot distinguish between different types of pathogen. By contrast, leukocytes involved in adaptive immunity are specific to different pathogens.
- Some adaptive immune system leukocytes have a ‘memory’. This leads to more effective immune responses with each exposure to the pathogen.
Congratulations on completing the course!
Now you’ve completed the course we would again appreciate a few minutes of your time to tell us a bit about your experience of studying it and what you plan to do next. We will use this information to provide better online experiences for all our learners and to share our findings with others. If you’d like to help, please fill in this optional survey .
If you enjoyed the course and are thinking about studying the module this material was adapted from (SDK100 Science and health, an evidence based approach) you can read more about the module and the health sciences qualification. You can also undertake a self-diagnostic quiz to check whether you have the necessary background knowledge and skills to cope with studying SDK100.
Take the next step

If you enjoyed this course, why not explore the subject further with our paid-for short course, Science: human genetics and health issues?
Find out more about Science: human genetics and health issues.
References
Acknowledgements
This unit was written by Basiro Davey, Carol Midgley, Claire Rostron and Daniel Berwick.
Except for third party materials and otherwise stated in the acknowledgements section, this content is made available under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 Licence.
The material acknowledged below is Proprietary and used under licence (not subject to Creative Commons Licence). Grateful acknowledgement is made to the following sources for permission to reproduce material in this unit:
Figure 8: Chris Knapton/Science Photo Library/Universal Images Group
Figure 12: Saturn Stills/Science Photo Library/Universal Images Group
Figure 14: The Global Polio Eradication Initiative © Copyright 2010
Every effort has been made to contact copyright owners. If any have been inadvertently overlooked, the publishers will be pleased to make the necessary arrangements at the first opportunity.
Don't miss out
If reading this text has inspired you to learn more, you may be interested in joining the millions of people who discover our free learning resources and qualifications by visiting The Open University – www.open.edu/ openlearn/ free-courses.
Copyright © 2015 The Open University