5.2 Energy sources in torpor and hibernation
For animals that show daily torpor, such as Siberian hamsters (Phodopus sungorus) and Djungarian hamsters (Phodopus campbelli) (Figure 28), blood glucose remains the respiratory fuel for several hours following its onset. Thereafter there is a gradual reduction in respiratory quotient (RQ) indicating a change to lipid metabolism as the metabolic rate is reduced.
There are some exceptions to the rule that lipids are the energy source of hibernation. Arctic ground squirrels (Spermophilus parryii) overwinter in hibernacula at temperatures that are substantially below freezing, whilst maintaining a T b at or just below 0° C. In laboratory studies, an increase in RQ from 0.71 towards 0.85–0.90, together with a fall in the amount of stored glycogen in liver and muscle, has been measured on entry to hibernation. Together, these changes point to a switch to glucose as the principal energy source.
Question 13
Why might carbohydrates be required as fuels in some hibernators?
Answer
First, following arousal episodes, glucose-utilizing tissues such as brain and blood cells would have a specific energy requirement that cannot be met by stored lipids. Secondly, there is a need to replenish lipids within BAT for the substantial needs of NST at the next arousal episode.
The latter need is not met from stored glycogen, but from gluconeogenesis, the biosynthesis of glucose from amino acids. The provision of amino acids requires the breakdown of muscle protein that contributes to the weight loss seen in many hibernators. The pattern of hibernation is frequently related to the combination of lipids with protein as the prime metabolic fuels. Black-tailed prairie dogs (Cynomys ludovicianus; Figure 7), whose diet is rich in polyunsaturated fatty acids (PUFA) and which use protein as a source of energy in the winter only during periods of reproductive activity, enter shallow torpor only infrequently and do not hibernate continuously between autumn and spring. However, the duration of torpor and size of the reduction of T b during hibernation can be increased by increasing the dietary intake of PUFA during the period before entry.
Arctic ground squirrels are capable of cooling their T b to −3° C, while simultaneously keeping the parts of the body involved in regulation and maintaining energy metabolism – the brain and intercapsular BAT – above zero. This ability shows that metabolic rate is regulated independently of body temperature, a conclusion which is further borne out by the data in Figure 29 (Buck and Barnes, 2000).
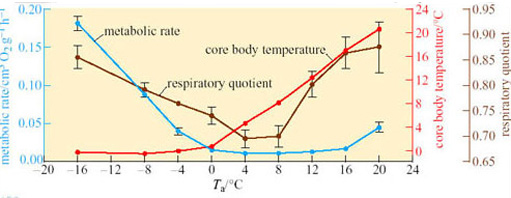
Between T a values of −16° C and 0, T b remains relatively constant but metabolic rate rises over 15-fold as the difference between T a and T b increases. At T a 0 to 20° C, T b increases with T a; however, metabolic rate does not change significantly from a T b of 0° C up to a critical temperature of 12° C, indicating the existence of a temperature-independent inhibition of metabolic rate.
Question 14
How do you interpret the change in respiratory quotient over the range of T a used in the investigation shown in Figure 29?
Answer
For most mammals, hibernation requires a shift away from the oxidation of carbohydrates and towards the oxidation of fatty acids released from stored triacylglycerols as the primary source of energy during torpor (see Box 1).
In 13-lined ground squirrels, the activity of PDK4 is elevated between three and eight-fold by increased translation of specific mRNA in three tissues important in energy metabolism – heart, skeletal muscle and WAT – during entry to torpor. It is important to bear in mind that translation of this gene, as well as that of a small number of other gene products (see Section 4), is selectively increased against a background of greatly reduced translational activity. This observation also points to the possibility of tissue-specific differences in the levels of biosynthesis of metabolically critical proteins.

