3.3.1 Dormancy in black and brown bears
The dormant state of bears differs from true hibernation in that the body temperature does not fall below 31–35° C and a major disturbance (such as an intruding biologist) can arouse them to full activity in a few minutes. Dormant bears do not eat, drink, urinate or defaecate, the heart rate drops from 50–60 beats min−1 to 8–12 beats min−1, and oxygen consumption is only 32% of that of actively foraging bears. Nonetheless, the rate of protein turnover, as measured by the rate of dilution of 14C-labelled amino acids injected into the blood, is three to five times higher during dormancy than in normal activity. Protein synthesis, particularly of enzymes involved in lipid and protein metabolism, also continues unabated during dormancy. The degradation of proteins to urea, however, is greatly slowed in dormancy. In these respects, the reciprocal changes in protein metabolism of the hibernating bear resemble those of humans and other mammals of tropical origin living on protein-deficient diets: essential amino acids are incorporated into proteins in the liver, but oxidation of amino acids and excretion of nitrogen are greatly reduced.
A small but significant quantity of urea is produced throughout dormancy but it is not excreted. Instead, it passes across the gut epithelium and into the lumen, where it is further degraded to ammonia (NH3) and carbon dioxide by the gut bacteria. The carbon dioxide is excreted with the respiratory gases, but the fate of the highly soluble, and in high concentrations toxic, ammonium ions (NH4+) is more interesting. In dormant bears, the blood concentrations of amino acids, total protein, urea and uric acid during dormancy are similar to those of active bears that are feeding regularly. Since there is almost no net elimination of the nitrogen, it must be re-incorporated into amino acids. The most important source of carbon for this process is glycerol. If 14C-labelled glycerol is injected into a dormant bear, the label quickly appears in alanine, then in other amino acids, and finally in plasma proteins.
SAQ 9
Where would the glycerol come from normally in a dormant bear?
Answer
Glycerol is produced from lipolysis of triacylglycerols.
The fatty acids released by lipolysis are used in energy metabolism, but much of the glycerol (that in other mammals is mainly oxidized) is combined with ammonia to form amino acids, which are incorporated into proteins in the normal way. This mechanism recycles the nitrogen so efficiently that the concentration of urea in the blood actually decreases slightly after several weeks of dormancy.
The rate of excretion of nitrogen can be estimated as the ratio of the concentrations of urea (U) to creatinine (C) in the blood (U:C ratio). Malcolm Ramsay and colleagues measured the U:C ratio in blood samples collected from wild polar bears in northern Canada (Ramsay et al., 1991). Creatinine is formed from the breakdown in muscle of phosphocreatine, a high energy phosphate compound, and is a minor but constant source of excreted nitrogen. In bears, the concentration of creatinine in blood plasma increased about threefold during the first 1–2 days in dormancy and then remained constant. The U:C ratio is around 50 in most mammals, especially carnivores that are eating regularly, and does not normally fall lower than 25, even during prolonged fasting. But U:C ratios of less than 10 are frequently measured in dormant black bears, indicating that during dormancy a high proportion of the urea is re-incorporated into proteins instead of being excreted (Nelson et al., 1984). Consequently, the bears’ lean body mass is hardly diminished even after months of dormancy and their muscle strength is unimpaired.
In starving humans and most other fasting animals, β-hydroxybutyrate and acetoacetate (ketone bodies) are formed by partial oxidation of fatty acids (see Figure 11). They are normally eliminated by further oxidation, but sometimes the presence of a high concentration of ketones disturbs the acid-base buffering of the blood and a comatose state called ketosis develops. In many hibernatory mammals, very high concentrations of ketone bodies trigger arousal. Ketone bodies increase in dormant bears as well but only to a maximum of ninefold between normal activity and dormancy and the toxic effects of ketosis have never been observed. Experiments in which labelled glycerol is injected into the blood of dormant bears show that, as well as being incorporated into amino acids, substantial amounts of labelled glycerol also appear in triacylglycerols.
SAQ 10
What does this observation show?
Answer
It shows that, as well as lipolysis of lipids stored in adipose tissue, resynthesis of triacylglycerols from fatty acids and glycerol is also occurring at a significant rate. The rate of triacylglycerol turnover may be higher during dormancy than during normal activity, and may limit the rate at which free fatty acids can enter the pathways that produce β-hydroxybutyrate and acetoacetate, thereby preventing ketosis and enabling the bears to sleep undisturbed for long periods.
SAQ 11
Are there any other metabolic advantages of utilizing fat during dormancy?
Answer
Oxidation of fat produces water. Since the bears do not drink during dormancy (except perhaps the occasional mouthful of snow), and the surrounding air is very dry, such metabolic water probably makes a significant contribution to water balance. Total body water, blood volume and the water content of red cells and plasma remain normal during dormancy, indicating that the water generated by such metabolism is indeed sufficient to offset the small losses due to respiration of the dry, cold air. Thus the large quantities of adipose tissue triacylglycerols in bears are much more than just an energy store: they are central to the bears’ metabolic adaptations to dormancy.
Measurements of composition of the respiratory gases reveal that the respiratory exchange ratio (RER) falls from 0.78 when the bears are fully active to 0.62–0.69 during dormancy. Such values are exceptionally low: the normal minimum RER for mammals, representing oxidation of lipid only, is 0.71. The low RER shows that some of the carbon dioxide that would normally be excreted through the lungs fails to appear. Carbon dioxide cannot be stored in significant quantities because as hydrogen carbonate (HCO3–), it alters the acid-base balance of body fluids, so it must be converted into non-volatile compounds, possibly by the microbes in the gut or by enzymes in the bear's liver.
Like other metabolic processes, the urea cycle and protein synthesis generate quite a lot of heat. The high rate of these processes during dormancy, together with the bears’ large size and thick, insulating fur, combine to maintain a much higher body temperature than that of small mammals in deep hibernation. Fully functional brown adipose tissue has not been demonstrated in bears, even in neonates, although small areas of white adipose tissue have some structural features that resemble those of BAT. Nonetheless, at a body mass of less than 1 kg, bears are smaller at birth, relative to the size of their parents, than any other eutherian mammal, and they are born in mid-winter or early spring.
Bears become fully active, eating and able to deal with predators within hours of leaving the den after weeks of dormancy. Astronauts after long periods in space and people recovering from illness or injury cannot do the same: especially in the elderly, the skeleton is weakened by more than a few days of immobilization as bone is reabsorbed. Vertebrae or limb bones may fracture under very weak forces; sometimes just standing is sufficient to cause injury. Measurements on people resuming activity after a period of bedrest show that the rate of reformation of bone is 2–3 times slower than its loss during immobilization. How to bears manage to avoid similar problems during and immediately after dormancy?
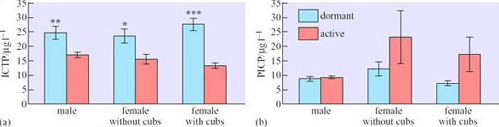
Carboxy-terminal propeptide of type 1 collagen (PICP) is the remnant of nascent type 1 collagen (the principal protein in bone) that is cleaved off by proteases as the protein is incorporated into bone. It can be measured in the blood serum, and thus serves as a convenient marker of bone formation. Another serum protein, carboxy-terminal cross-linked telopeptide of type 1 procollagen (ICTP), acts as a marker of bone resorption. Figure 13 shows some measurements of these proteins in breeding and non-breeding black bears (Ursus americanus) during dormancy and when actively feeding in the wild in the mountainous region of Virginia, USA (Donahue et al., 2003).
SAQ 12
What conclusions about bone loss and reformation can you draw from Figure 13?
Answer
No statistically significant differences between dormant and active bears were found in the measurements of PICP, though there was a trend towards higher values for active females. In contrast, ICTP, especially in females who were feeding cubs, was higher while they were in the den.
SAQ 13
Do the data in Figure 13 explain how bears avoid increased risk of bone fractures just after emerging from the den after a period of dormancy?
Answer
No, not fully. Bone loss (Figure 13a) is still higher during dormancy (though the differences between measurements from bears found in dens and full activity are not as great as between sedentary and exercising people or rats). But the data provide no evidence that bone formation is consistently greater while the bears are active.
SAQ 14
Could the investigation have failed to reveal important aspects of the time course of bone turnover during dormancy and activity?
Answer
Yes. This investigation would fail to detect any brief period of acceleration of bone formation just before or just after emergence from the den. It is important to study the markers of bone turnover during the transitions between dormancy and activity.
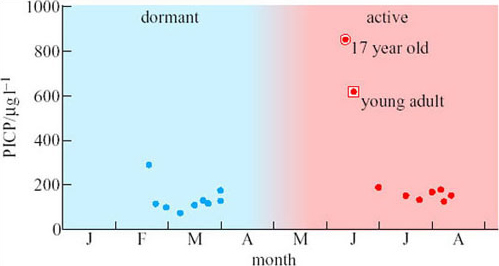
The scientists were able to obtain measurements of the bone formation marker (PICP) before, during and after emergence from a few bears (Figure 14). These measurements show that PICP is four- to fivefold higher early in the remobilization period compared with mid-summer. This brief peak in the concentration of PICP may indicate that bone formation increases just when it is most needed.
SAQ 15
Do the data in Figure 14 indicate exactly how long the peak concentration of PICP in bears emerging from dormancy lasts?
Answer
No. Unfortunately, no bears were found and tranquilized between the end of March and the beginning of June, so we have no information about this crucial period. Such are the problems of studying wild animals under natural conditions.
This more efficient compensatory mechanism for recovering from immobilization-induced bone loss, combined with low bone loss during disuse, enables bears to switch abruptly between dormancy and full activity. The control mechanisms that prompt the rapid increase in bone formation remain to be explored. Its elucidation might enable physicians to induce similar effects in people recovering from hip-replacement operations and other situations in which long periods of immobilization are unavoidable.
SAQ 16
Why are the differences between the values for dormancy and activity greatest for females that are suckling cubs (Figure 13)?
Answer
Lactating females are transferring some of the minerals (especially calcium) in the bones to the milk (where it is incorporated into the cubs’ skeletons), further weakening their own skeleton. Breeding females thus need to maximize bone reformation after dormancy.
