2.3 Behavioural strategies of evaders
Small animals, classified as evaders, include desert amphibians and reptiles, and also mammals, rodents and insectivores. The term ‘evaders’ refers to the animals’ behaviour, which helps to prevent overheating of the body on hot sunny days, and avoids the need for cooling by evaporative water loss, which is not feasible for small animals living in an arid habitat. Evaders make use of microenvironments such as shady rock crevices, underground burrows and shade cast by plants, for behavioural thermoregulation. Evaders also prevent excessive cooling of the body by behaviour, retreating to shelter when T a plummets at night.
The ultimate evaders are desert frogs such as Cyclorana spp. (Figure 10) and Neobatrachus spp. (Figure 11) from Australia, which spend most of the year in aestivation, inside a burrow. Aestivation is a special kind of dormancy, which enables animals to survive lack of water and high T a during a hot dry season. During the short rainy season, desert frogs accumulate water in the bladder, where it remains during aestivation. A famous example, Cyclorana platycephala (Figure 10), is known as the water-holding frog; aboriginal people used to dig up the aestivating frogs and squeeze them, in order to collect and drink the water.


During aestivation, the frogs are protected from losing water to the dry soil in the burrow by a cocoon. At the end of the rainy season, the frogs burrow into the soil, and the skin undergoes a type of moulting process in which layers of epidermis are separated from the body but not shed, forming a protective cocoon, covering all parts of the body apart from the nostril openings. The cocoon thickens, becoming heavily keratinised, and prevents loss of water from the frog's body during the 9–10 months of aestivation. At the start of the rainy season, heavy rain with consequent seepage of water into the frogs' burrows, stimulates the frogs to emerge. Breeding and feeding occur during the short wet season.
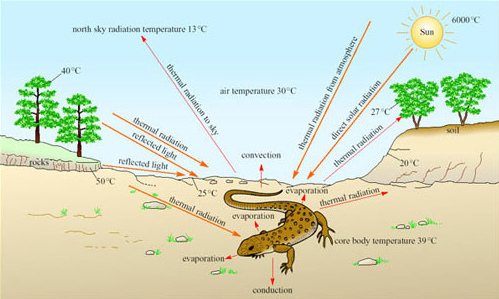
Reptiles with a scaly keratinised skin are not so prone to evaporative water loss as amphibians, and are the vertebrates that you are most likely to see on a visit to a desert. Reptiles are ectotherms and rely on solar radiation for warming the body, and maintaining high T b during the day. Desert reptiles have no problem in gaining heat for maintaining T b at a high level on hot sunny days (Figure 12).
Activity 1
What are the sources of energy gain and routes of heat loss for the lizard?
Answer
The lizard gains heat energy via thermal radiation from the Sun, the atmosphere and the ground. Heat energy is lost via conduction from the body to the ground, by evaporative water loss, convection and thermal radiation to the sky.
On a hot sunny day, more heat is gained than lost, and it is important for a desert reptile to avoid overheating. It is equally important to reduce loss of body heat when T a plummets at night or during the winter.
During the day, reptiles may move between warm and cool areas in order to maintain T b. This movement between warm and cool areas for maintaining eccritic temperature is called shuttling. Those species that maintain high stable T b when environmental conditions allow by adopting heliothermic strategies, are called thermal specialists. In contrast, there are some species, known as thermal generalists, which allow their T b to fluctuate and decline, even when they could shuttle between sun and shade to maintain high stable T b during the day, or use their burrow at night to prevent cooling of T b to the outside T a. Bedriagai's skink (Chalcides bedriagai; Figure 13) is a thermal generalist, preferring to spend a lot of time hiding under rocks rather than basking in the sun.

The side-blotched lizard (Uta stansburiana; Figure 14), found in the Sonoran desert, is a typical thermal specialist. It is a small species, only 4–6 cm long when full grown.

In the morning, Uta warms by basking, initially orientating itself at right angles to the Sun's rays and flattening the body against the substratum for maximum exposure to solar radiation. When warmed Uta turns the body so that it faces the Sun while resting. Uta maintains T b around 36–38°C. Active foraging for insects, scorpions and spiders may overheat the body, and for cooling off, especially around noon, Uta moves to the shade of rocks and scrubby bushes. Shuttling in this way enables this species to stay active during the day for most of the year except in areas where winter temperatures dip to freezing.
A few desert reptiles are nocturnal; the Moorish gecko (Tarentola mauretanica; Figure 15), is found in arid regions in North Africa (also in Spain, France and Greece, so it is not restricted to deserts).

Tarentola is most active for a few hours after sunset. During the night, its T b is as low as 18°C, and can fluctuate by up to 11°C. Recall that lizards that tolerate wide fluctuations in T b, even when they could use features of the environment to maintain a steady T b, are known as thermal generalists. The Moorish gecko is a thermal generalist at night, when it is active rather than resting in its burrow. During the early morning the Moorish gecko basks in the sunlight and its skin darkens until almost black. At night the gecko is very pale.
Activity 2
What advantages do the changes in skin colour give?
Answer
Dark colours absorb and radiate heat better than light colours. At night a light colour should reduce heat loss by radiation, and there is not much heat available to absorb. During the day, dark skin promotes absorption of solar heat. Although radiation to the atmosphere by the dark skin is also promoted, the energy so lost is of little significance compared to the large amount of solar heat absorbed.
The advantage to the gecko of warming up in the morning is uncertain, but it is possible that a physiological process such as digestion of the food eaten during the night requires a higher T b than the gecko can maintain at night.
The ability of the gecko to vary skin colour shows that behavioural thermoregulation in reptiles is supplemented by physiological mechanisms, which we will explore further in Section 3.4.
Sheltering in the available shade in the desert, or being active at night, are simple strategies for keeping T b below lethal levels. In sandy desert areas, the sand itself plays an important role in behavioural thermoregulatory strategies. The Mojave fringe-toed lizard (Uma scoparia) (Figure 16) is restricted to fine, wind-blown sand, e.g. in dunes, dry lake beds and desert scrub in the Mojave desert. Burrows in sand collapse immediately or soon after the animal has moved on, so animals buried in sand rely on air trapped between sand particles for breathing. Uma is a ‘sand-swimmer’ and its dorsoventrally flattened body and shovel-shaped head facilitate movement through the sand, which is especially important when escaping from predators such as snakes and badgers.

The eyelids are protected from sand by large eyelid fringe scales. The digits have large lamellar fringes, elongated scales, especially long on the hind feet, which enable the lizards to run at speed on the sand surface. Uma grows up to about 110 mm in length, and its activity pattern is diurnal, varying according to ambient temperature. In March and April Uma is active for short periods because of the low spring temperatures in the Mojave. In summer, from May to September, the lizards are active during mornings and late afternoons, feeding on insects and plants. Sand-swimming lizards are also found in the Namib desert and include the wedge-snouted sand lizard (Meroles cuneirostris).
The data in Figure 17 were collected from a sand dune slope in the Namib desert.
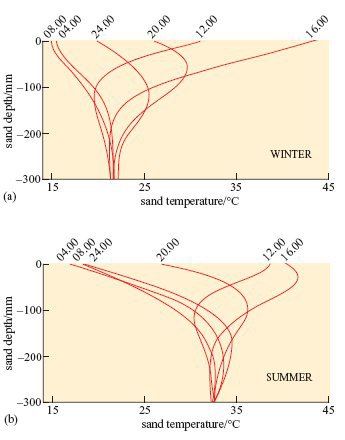
Although the temperatures of sand at various depths in the Mojave desert would not be precisely the same as those in the Namib, the physical characteristics and thermal environment provided by dry sand are broadly the same in all deserts at similar T a.
A benign temperature is available below the surface at all times of the day in both seasons, in spite of extremes on the surface. These surface temperature extremes are not very different in summer and winter. The high afternoon surface temperature in winter is due to hot, dry winds (Berg winds) that reach the desert in the winter months.
Activity 3
Examine the data in Figure 17 and suggest the advantages for a sand-swimming lizard of the following strategies:
The lizard ‘swims’ down to 60 mm depth at 12.00 hours in summer, when surface temperatures can reach 40°C or more.
In winter, the sand-swimmer remains in a state of dormancy for a month at 300 mm depth in the sand, when surface temperature can occasionally drop below freezing at night.
Answer
At 12.00 hours, when T a is 40°C at the surface, by burrowing to a depth of 60 mm the lizard reaches a microenvironment where T a is significantly lower, about 32°C (Figure 17b). The lizard loses body heat by conduction and thereby avoids a dangerous increase in body temperature.
In winter when ambient temperatures can drop to below freezing, the temperature at 300 mm depth remains constant at around 21°C (Figure 17a). The lizard thereby avoids low T a at the surface and is not at risk of freezing when T a drops to
Burrows provide important microenvironments for many desert evaders, and their structure and use vary between species. The desert tortoise (Xerobates agassizii; Figure 18) lives in deserts in the USA and Mexico, and feeds on annual herbs, cacti and shrubs, obtaining most of its water from the plants.

In the Mojave desert, the tortoises live in sandy areas as well as rocky hillsides, including scrub-type vegetation, joshua tree/yucca and creosote bush/ocatillo habitats. For the tortoises, burrows are important refuges for thermoregulation, summer aestivation and winter hibernation. Tortoise burrows in the Mojave desert are extensive and can be up to 12 m long; the same burrows are used for many generations, and are shared with other species such as burrowing owls and ground squirrels. Each desert tortoise may use up to 12 burrows in its home range and each burrow is used by different tortoises at different times. For short rest periods during the day tortoises dig shallow depressions, known as pallets, which barely cover the carapace.
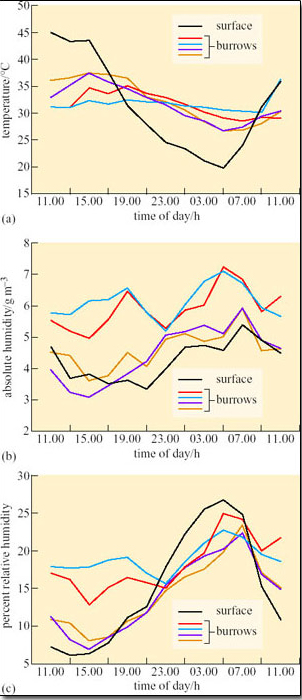
Susan Bulova (2002) compared temperature and humidity in four unoccupied desert tortoise burrows, and the surface over 24 hours on a summer day in the Mojave desert (Figure 19a–c).
Activity 4
Compare the fluctuations in T a (Figure 19a), in the burrows and on the surface.
Compare the fluctuations in absolute humidity (a.h.) and relative humidity (r.h.) (Figures 19b and c) in the burrows and on the surface.
Answer
T a inside each of the burrows fluctuated by about 2–10°C, ranging from about 27–37°C. Surface T a fluctuated from a low of 20°C at 05.00 h to 45°C at 11.00 h. Compared to surface T a, burrow T a remained relatively stable, being cooler than surface T a from 09.00 h–17.00 h and warmer than surface T a from 17.00 h–09.00 h.
Absolute humidity was generally lower on the surface than inside the burrows, but for two of the burrows measured a.h. values were always close to surface a.h. values. Relative humidity was higher in the burrow than on the surface from 09.00 h to 21.00 h but two of the burrows had r.h. values quite close to surface r.h. from 15.00 h to 23.00 h.
Bear in mind that when occupied by a tortoise, a burrow's relative humidity may rise to 40 per cent because of the tortoise's water loss by evaporation from the lungs, exposed skin and eyes. Stable T a and humidity in the burrow protect the tortoise from extremes of high T a and from winter frosts. Bulova noticed that tortoises are fussy about the burrow selected for resting. At the end of foraging, tortoises were observed to enter and leave several burrows before settling. Mojave desert tortoises are active between March and June, a time when the winter rains have stimulated the growth of annual plants, providing abundant food for the tortoises. The tortoises begin foraging during the morning but usually by noon they have moved into pallets and burrows to shelter from high T a. At night, burrows provide shelter from low T a and also protection from nocturnal predators such as kit foxes and badgers. By the end of June, when surface temperature may reach 60°C, and annual plants have dried up, the tortoises retreat to their deep burrows and aestivate, a behaviour that helps to conserve body water. During aestivation, up to a quarter of the tortoises' body mass may be water stored in the bladder. Occasionally an aestivating tortoise emerges to drink during summer thunderstorms. In the eastern Mojave desert tortoises are active for most of the summer because there, summer rainstorms provide sufficient new plant growth. For their winter hibernation, tortoises aggregate in the burrows; up to 25 individuals have been found in one burrow. Hibernation lasts from October to the end of February, and during this time T b of the tortoises is the same temperature as the burrow, around 5–16°C in winter. Note therefore that hibernation in the desert tortoise is not the same physiological process as it is in hibernating mammals . Reptiles do not regulate T b physiologically during hibernation; T b is the same as burrow T a. You will find that in some references, reptile ‘hibernation’ is termed ‘brumation’.
You may be surprised to learn that like desert ectotherms, small desert rodents also depend on burrows for thermoregulation. Merriam's kangaroo rat (Dipodomys merriami; Figure 20) is a typical evader, living in the Sonoran desert, Arizona, and in Death Valley, California, one of the hottest and driest areas in the Western Hemisphere.

Individuals live in a maze of burrows, which they defend. They remain in their burrows during the day, and often plug the entrance with soil. At night kangaroo rats emerge from their burrows for just two hours to collect seeds, in particular seeds of the creosote bush, which they push into their cheek pouches, returning at intervals to empty the food into their burrow. In this way, food caches are built up; kangaroo rats always eat inside the burrow, drawing on their food cache. Inside the burrow, the air is cooler and more humid than above the ground, as moisture from respiratory water loss accumulates. Measurements made on similar burrows in the Negev desert, Israel, showed T a of around 26°C at 1 metre depth for 24 hours per day when ambient temperature above ground ranged from 16–44°C. However, not all small desert animals can burrow.
The desert wood rat (Neotoma lepida) lives in deserts in the southern USA, including Death Valley, California. Wood rats do not burrow but build elaborate houses around the base of cacti or shrubs, amongst a patch of agaves, or beneath a rock outcrop. Wood rat houses can reach huge sizes and their interior is significantly cooler, by about 5°C, than the outside during the heat of the day. Desert wood rats shelter in their houses during the day, and emerge to forage at night, eating creosote bush, cholla, prickly pear cactus and agave.