1 How much energy is in food?
The top of the nutritional information on a food label usually shows you how much energy the food contains. It will be given in both joules and calories. These are just two different ways of measuring energy – similar to how you can measure length in either inches or centimetres.
The unit of energy is named after James Prescott Joule, who was born in Salford, Lancashire in 1818. As an adult, he took over from his father as manager of the family brewery. Science was just a hobby.
Joule was fascinated by different types of energy and how one could be converted to another. Particularly puzzling was how the energy generated by, say, a falling weight could be converted to heat.
On his honeymoon in the Swiss Alps, Joule took a very accurate thermometer with him to measure the temperature of the water at the top and bottom of a waterfall. He wanted to work out how much heat was generated by the falling water. (There is no record of what his new wife Amelia thought about his honeymoon activities.)
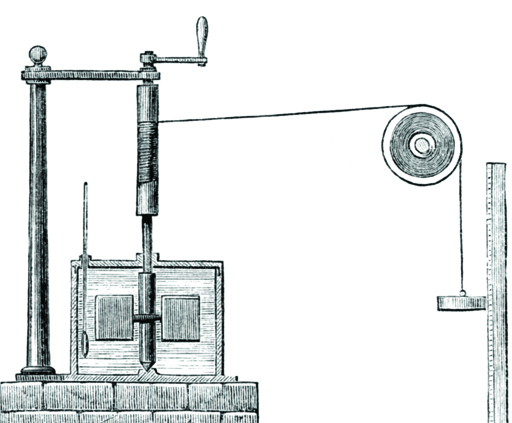
Figure 3 shows the apparatus Joule built for one of his other experiments. The ‘box’ on the left contains water and a paddle that is turned by the falling weight and a thermometer measures the temperature rise of the water.
In 1889, Joule died and on his tombstone the number 772.55 is engraved. This was the value he calculated for the energy in foot-pounds that would cause a temperature rise in one pound of water from 60 to 61 degrees Fahrenheit. One foot-pound is the energy produced when one pound in weight falls by a distance of one foot.
When the modern SI system of naming units was formalised, pounds were replaced by kilograms and feet by metres. The equivalent unit of energy was called a joule, after this great scientist.
One joule is a very small amount of energy. It is approximately the energy released when a small apple falls one metre to the ground. So, generally we work with the unit of a kilojoule (one thousand joules). One kilojoule (kJ) would heat one kilogram (one litre) of water by 0.24 °C.
Also on the food label, and commonly used when discussing food and nutrition, is the energy value in kilocalories (kcal). Sometimes, you may see the unit that should correctly be called a kilocalorie is instead called a Calorie, with a capital C. When people talk about how many ‘calories’ there are in food, they generally mean kilocalories.
Despite the calorie not being an SI unit, it has remained in common usage. It was first defined by Nicolas Clément (1779–1841), who lived in France at about the same time as Joule. The name is derived from the Latin word calor, meaning ‘heat’. It has the advantage of being rather convenient in experimental situations.