2.7.1 Adipose tissue
In this course, there have been several references to adipose tissue. You probably have a sense of a rather inert (and, perhaps, hard to shift!) tissue acting as a long-term energy store, but this is only a small part of the story.
Adipose tissue consists of individual fat cells (adipocytes (Figure 12)) together with connective tissue, a rich blood supply that is shared with adjacent tissues, and innervation from the sympathetic nervous system. The adipocytes are large, clear cells containing stored lipids. The volume of individual adipocytes varies markedly, perhaps as much as tenfold, as adult humans become more or less obese. Mature adipocytes do not divide but differentiate from very much smaller cells known as pre-adipocytes. The number of adipocytes in an adult is influenced by nutrition in fetal and pre-adult life.
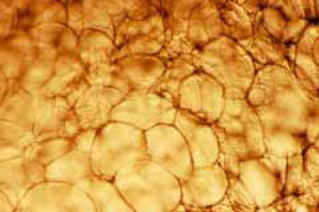
Adipocytes are often concentrated into fat depots which are characteristic of both species (e.g. a camel's humps) and gender (e.g. a human female's breasts). However, subcutaneous, intramuscular and intra-abdominal fat depots are found in most mammals. The paunch, characteristic of many primates, including our own species, is formed from a thickening of the outer wall of the abdomen. These various fat depots show differential tendencies to increase in size as an individual becomes more obese. For example, more especially in human males, the paunch and subcutaneous fat in the thigh may be greatly increased, whereas as the intramuscular fat in the calf hardly changes in quantity (Figure 13).
Adipose tissue is usually seen as having a number of functions. You already know that the production of leptin by adipose tissue is a critical component of the homeostatic control of energy balance. In the longer term, adipose tissue may represent a significant energy store to be used in times of low energy availability. In humans, and other mammals, it is also known that release of fatty acids from adipose tissue is an important source of energy during intense or protracted exercise. Interestingly it turns out that there are substantial differences in the ability of different adipose tissue depots to release fatty acids at such times. Much of the release comes from subcutaneous fat rather than, for example, fat in the abdominal paunch. Raised levels of adrenalin in the blood stream together with release of noradrenalin from the sympathetic nerve endings within adipose tissue triggers this process.
It is also increasingly clear that adipose tissue plays a critical role in the homeostasis of fatty acid levels in other body tissues such as the muscles and the liver. This insight has come, in part, from the study of individuals who have abnormally low levels of adipose tissue, a condition known as congenital lipodystrophy. An example is the rare Berardinelli-Seip syndrome which results from a mutation in one of several genes coding for the enzymes critical to the synthesis of triacylglycerols from fatty acids.
Activity 30
Why would an inability to synthesize triacylglycerols impair the ability to store fat in adipose tissue?
Answer
Triacylglycerols are the form in which fat is stored within adipose tissue. Triacylglycerols are synthesized from glycerol and the fatty acids that result from digestion of fats in the diet. A loss of the synthetic pathway for triacylglycerols prevents adipose tissue from storing fat, and the consequence during development is that very little adipose tissue is produced.
One result of congenital lipodystrophy is that high levels of fatty acids accumulate in both muscle and liver. Ironically, people with lipodystrophy share many physiological characteristics with those who are obese. In particular they develop insulin resistance and type 2 diabetes by early puberty. This occurs because the high levels of fatty acids in muscle and other internal organs disturb signalling initiated by the binding of insulin to the insulin receptor. Of course, another characteristic of those with lipodystrophy, but one that distinguishes them from obese individuals, is that they have very low circulating levels of the hormone leptin.
Activity 31
What are the likely consequences of low leptin levels in these individuals and how might they be treated?
Answer
The low levels of leptin will lead to enhanced appetite as a consequence of increased NPY and decreased -MSH levels in the hypothalamus. The increased food intake will simply exacerbate the increased levels of fatty acids within muscle and liver. One potential treatment would be to administer leptin. Early indications (2004) are that this strategy may be successful.
A further function of one specialized form of adipose tissue is heat generation from metabolism of fat. Most adipose tissue has a white appearance, but one specialized form is light brown in appearance. The brown colouration arises from a high concentration of mitochondria within these adipocytes. The normal function of mitochondria is to produce ATP, which is used to provide the chemical energy necessary for the synthesis of complex molecules, such as protein, and many other processes vital to the maintenance and growth of cells. However, in the mitochondria within brown adipose tissue the usual production of chemical energy in the form of ATP can be uncoupled from the oxidative breakdown of fatty acids, sugars and amino acids shown in Figure 3. Since a basic law of thermodynamics states that energy cannot be lost, but only transformed from one form to another, the chemical energy that would have been stored in the ATP must appear somewhere else. In fact it is released from the cell in the form of heat. This uncoupling can be switched on by the release of noradrenalin from the sympathetic nerves that innervate brown adipose tissue. Heat generated by brown adipose tissue metabolism is of particular importance in infants who have a greater surface area to volume ratio than have adults, and also in the process of cold adaptation. At one time it was thought that the stimulation of heat generation by brown adipose tissue metabolism might produce a novel treatment of obesity in which, analogous to regulation of fluid balance by the kidney, energy balance could be achieved by increasing energy loss rather than by restricting energy intake. However, it remains unclear whether adult humans retain enough brown adipose tissue to make a real difference to total energy balance. In addition, it has not proved an easy matter to design drugs that act selectively at the noradrenergic receptors in adipose tissue without having unacceptable side effects elsewhere in the body.
In the same way, as a study described by Blundell et al. (2003) makes clear, exhortation to exercise may also not be very fruitful. In a four-week experiment, both normal and overweight women were prescribed the levels of exercise recommended by the World Health Organization (20 minutes aerobic activity 3–5 times a week). However, there was little effect on either energy expenditure or body weight in the participants, although the prescribed level of activity would have been predicted to lead to a loss of more than 1 kg providing energy intake did not change. The authors reported that their participants found even these moderate levels of activity hard to achieve and speculated that individuals who are already vulnerable to obesity in later life are unlikely to follow a prescribed exercise programme without considerable additional support. Nevertheless, epidemiological data (e.g. Sternfeld et al., 2004) do suggest that even mild exercise over a prolonged period is associated with a worthwhile reduction in the normal age-related increase in body weight observed in middle-aged women.
Current drug treatments for obesity are limited, in part because of problems with side effects. One recent (2004) development, currently in late phase clinical trials, is a drug, known as rimonabant, which is an antagonist at the brain receptors for cannabinoid neurotransmitters. These are the same receptors that mediate the effects of tetrahydrocannabinol (THC), the most active component of marijuana (‘grass’ or ‘hash’) and they are found, amongst other places, in the ventral striatum (Figure 8). Interestingly, one side-effect of marijuana use, first reported more than 2000 years ago, is stimulation of appetite (‘the munchies’).
Activity 32
What effect would you expect a cannabinoid antagonist to have on food intake?
Answer
It should have the opposite effect to marijuana or THC and reduce food intake.
It may be that short term prescription of drugs similar to this, in combination with interventions that are aimed at longer term changes in an individual's diet and activity patterns, will provide more successful therapeutic options for the overweight and obese.
