3.5 Monomeric G proteins
We shall discuss monomeric G proteins (also called small G proteins or small GTPases) separately from the trimeric G proteins for three reasons: their upstream activators are different, they tend to have different target proteins, and they commonly operate within different signalling pathways.
What structural features and activities do monomeric G proteins share with trimeric G proteins?
You may have thought of the following:
a.Monomeric G proteins are structurally related to the α subunit of trimeric G proteins.
b.They bind GTP and hydrolyse it to GDP, acting as a molecular switch.
c.The rate of GTP hydrolysis can be increased by GAP proteins.
d.They are tethered to the internal surface of the plasma membrane by means of post-translational lipid modifications (Section 1.6).
There are many types of monomeric G proteins, involved in a myriad of cellular processes; examples include Ran, which has a role in the nuclear localization of proteins Rab, with a role in endocytosis or the Rho family, which functions in cell adhesion and migration. The best-studied monomeric G protein is Ras, which we shall consider here in some detail. Ras is structurally similar to the α subunit of trimeric G proteins. In fact, the GTP-binding domain of Gα is known as the Ras domain (Figure 35).
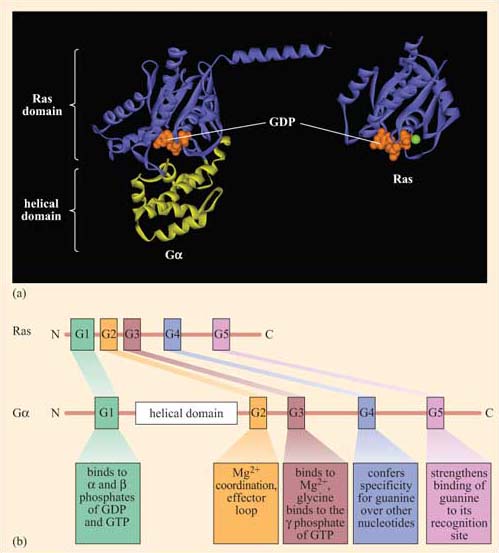
Ras forms part of the signal transduction pathway from most RTKs, including the growth factor receptors. As such, it is central to regulation of the growth, proliferation and differentiation of cells, and is involved in the formation of tumours when these pathways become dysfunctional. Ras homologues have been found to regulate vulval development in C. elegans and photoreceptor development in Drosophila, providing excellent experimental models in which to study its function in development. There are three members of the mammalian Ras family – H-Ras (first discovered in its viral form in Harvey murine sarcoma virus), K-Ras (from Kirsten sarcoma virus) and N-Ras (or neural Ras). There are also more than 70 other small G proteins, which share regions of sequence homology and are involved in a whole variety of cell processes.
For example, Rho and Rac have been shown to mediate changes to the actin cytoskeleton, leading to stress fibre formation and lamellipodia formation, respectively.
Ras is activated following stimulation of many RTKs (Figure 36). How does this happen? It is a much more indirect process than for trimeric G proteins, where conformational change in the receptor directly induces conformational change and activation of the G protein. Instead, what usually happens is firstly that autophosphorylation of RTKs creates docking sites for SH2-containing proteins. One of these SH2-containing proteins is an adaptor protein called Grb-2 (growth factor receptor-bound protein 2). Grb-2 consists of little more than two SH3 domains flanking an SH2 domain (Figure 13). Grb-2 binds to the activated receptor via its SH2 domain. Sometimes the RTK does not contain the correct docking sequences for Grb-2, in which case an intermediary adaptor protein such as Shc (another SH2 protein also in Figure 13) is used.
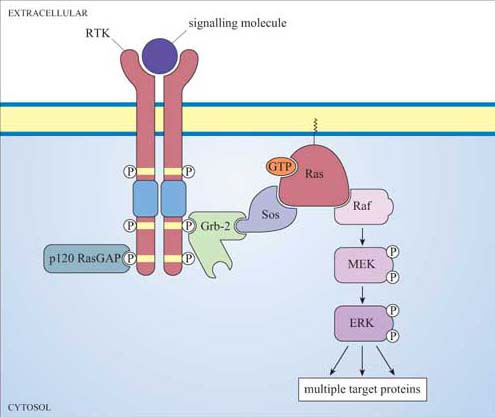
One of the SH3 domains of Grb-2 bind to a Ras activator protein, which contains the appropriate proline-rich domains. Ras activators are proteins that, by binding to Ras, stimulate Ras to exchange its GDP for a molecule of GTP.
What is the name for this class of protein?
Guanine nucleotide exchange factors, GEFs (Section 1.6).
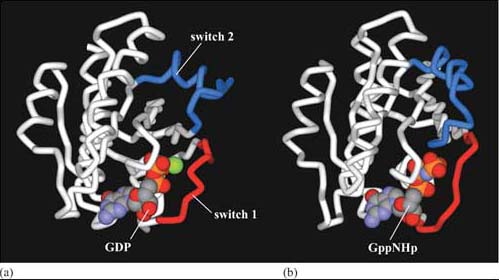
The usual GEF that acts on Ras is called Sos (Figure 36). You can thank Drosophila researchers for this name! The photoreceptor studied by them is called R7, and the gene for the tyrosine kinase receptor involved in photoreceptor development is called ‘sevenless’ (because sevenless mutants lack R7 photoreceptors). The GEF was discovered to be downstream of the receptor, and so its gene was called son of sevenless. Because Sos is recruited to the plasma membrane by Grb-2 following receptor activation, it is then in the right place to bind to Ras (which is permanently tethered there) and encourage it to bind GTP. Since GEFs promote GTP binding and RasGAPs promote hydrolysis of GTP to GDP, the overall model is that GEFs switch Ras on, and GAPs switch Ras off (look back at Figure 8b). GDP-bound Ras is in an inactive conformation, but when it is bound to GTP, two ‘switch’ regions (the main one of which is an α helix) are affected, resulting in the adoption of an active conformation (Figure 37). These switch regions are implicated in binding to effectors and also to RasGAP.
It is remarkable that such a small and permanently membrane-bound protein as Ras is able to mediate so many profound changes within a cell. However, we can see how it can serve as an integrator of signalling information via GEFs and GAPs. As for its effectors, Ras is known to affect PI 3-kinase, but it achieves most of its effects by activating the first component of a key signal transduction cascade called the MAP kinase pathway (Figure 36), which serves to amplify signals and operate over a rather longer time frame than, say, second messenger systems. Moreover, it has many different cellular targets, including transcription factors, which it activates.