2.4 Chain conformations
The repeat units or chains shown in the previous figures are all static representations of real chains and they are therefore of limited use. The key idea we need to explore real chains, and their influence on properties is that of the conformation of a chain. A single conformation is just a single shape that a chain can adopt, so that for example, when a polyethylene chain is shown as a linear zig-zag, this is one possible conformation. Static formulae do not show an important aspect of real chains – their oscillation and movement as a result of thermal vibrations of the molecular structure. These chain molecular motions increase in both frequency and amplitude as the temperature is raised, the most important being rotation about single bonds. By contrast, double bonds are rigid and the adjacent atoms are immobile. As a consequence of extra rotational motion, the number of conformations a chain can adopt increases rapidly with increasing temperature. The question then arises whether or not there are any conformations which are more stable than others at any particular temperature, given the chemical structure of the repeat unit.
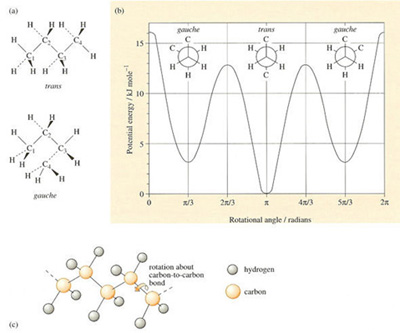
To answer the question, consider the simple molecule of n-butane (Table 4), which consists of four carbon atoms linked together in a linear chain together with hydrogen atoms along the periphery. There will be considerable interference between adjacent hydrogen atoms when they are aligned. The interference will be lower when they rotate about their own axis to give a staggered conformation (Figure 25 (a)). Concentrating on the two central carbon atoms (C2 and C3), and looking along the C2—C3 axis, it is also clear that there are two different ways in which the outer carbon atoms (C1 or C4) can be situated with respect to one another. One position where C1 and C4 are in opposite positions is known as the trans conformation, the other where they are adjacent is known as the gauche conformation. It will be readily apparent from the butane model that interference occurs between the hydrogen atoms on C1 and C4 carbon atoms in the gauche conformation. This so-called steric hindrance makes the gauche conformation of higher energy than the trans conformation, as shown in Figure 25(b). The net effect then is that the trans conformation is favoured and this also applies in longer chain hydrocarbons.
| Name | Formula | n | Tm/°C | Density of liquid/Mg m−3 | State at 20 °C |
|---|---|---|---|---|---|
| ethane | C2H6 | 1 | −172 | 0.5462 | gas |
| n-butane | C4H10 | 2 | −135 | 0.5788 | gas |
| n-octane | C8H18 | 4 | −57 | 0.7028 | liquid |
| n-hexadecane | C16H34 | 8 | 18 | 0.7749 | liquid |
| n-triacontane | C30H62 | 15 | 66 | 0.7786 | waxy solid |
| n-heptacontane | C70H142 | 35 | 105 | 0.7940 | waxy solid |
| polyethylene | C700H1402 | 350 | 130–140 | ca. 0.08 | solid polymer |
When polyethylene crystallises it adopts an overall linear conformation in which the carbon atoms form a planar zig-zag, where they are all trans with respect to their near neighbours. In the non-crystalline state, the relative proportion of trans and gauche conformations, Nt and Ng, can be predicted using Boltzmann's equation:
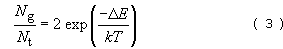
where ΔE is the net energy difference between the two states (≈3 kJ mole−1 in this instance), k is Boltzmann's constant and T the absolute temperature. The factor of two is necessary because two gauche states are possible. Since
it follows that, as the temperature rises, the energetic contribution decreases and so the right-hand term increases; this means that more and more gauche conformations are adopted as the temperature rises. The net result is that the randomly coiled polyethylene chain tends to contract in size with rise in temperature as linear zig-zag trans conformers become less likely along the chain.
Exercise 7
The viscosity of engine oil is very sensitive to changes in temperature, but can be made much less sensitive by adding a polymer whose chain expands as the temperature rises. One such additive is poly(ethylene oxide) (PEO), whose randomly coiled chain expands in size when the temperature is raised. Explain the effect in terms of rotation about single bonds in the main chain.
Answer
The effect must be the opposite of that in polyethylene, namely, the trans conformers have a higher energy than the gauche conformers. In other words, the linear zig-zag conformation formed by a succession of trans positions along the backbone chain becomes more favourable as the temperature is raised. The chain is now much longer than in a random coil conformation, so the viscosity would be expected to be higher.
