3.3.2 Benzene, toluene and xylene
In addition to benzene itself, the catalytic reformer also produces ethylbenzene, toluene and the isomeric xylenes directly. The demand for ethylbenzene is always great as a source of styrene monomer, but toluene does not find great use apart from a relatively small application in polyurethane. This is why most toluene is de-alkylated to increase overall benzene production. A similar problem exists with the xylenes:
Para-xylene is most widely used as a source of terephthalic acid for PET (Table 5), a blow moulding and fibre forming polymer. Although demand is lower, the ortho isomer is used to make phthalic anhydride of use in thermosetting resins and paints,
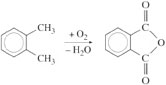
The unused meta- and ortho-xylenes are downgraded to a mixed solvent, a step which clearly represents a loss of added value. Some attempts have been made to use meta-substituted groups in polymers, Nomex fibre for example, but the demand for this speciality material is insufficient to exploit the amount of meta-xylene available from reformed naphtha.
A much more complex procedure is necessary to make the two monomers for nylon 6,6. Benzene must first be hydrogenated back to cyclohexane which then undergoes oxidation and ring scission to create hexamethyl-enediamine and adipic acid in five and four steps respectively. It is an irony of petrochemical processing that such a long-winded procedure is needed – and it is a direct consequence of the complex mixtures of hydrocarbons produced in the first major processes of refining.
