1.4.4 Inhibitors of metabolic reactions
Antibiotics that disrupt essential bacterial metabolic pathways are acting as antimetabolites. These chemicals are structurally similar to natural metabolites but just different enough to interfere with normal cell function.
For example, trimethoprim inhibits the synthesis of folic acid, a vitamin that bacteria, unlike humans, must make themselves. Trimethoprim is a structural analogue of dihydrofolic acid, an intermediate compound in the folic acid pathway. Trimethoprim out-competes dihydrofolic acid to react with a specific bacterial enzyme in the pathway, thereby interrupting folic acid synthesis and inhibiting bacterial growth (Figure 13a).

Figure 13a The folic acid pathway. Trimethoprim prevents the enzyme dihydrofolate reductase reacting with the intermediate compound dihydrofolic acid, thereby blocking the pathway at the point shown.
Show description|Hide descriptionThis figure shows the folic acid pathway. Para-aminobenzoic acid and pteridine are converted into dihydrpteroic acid by dihydorpteroate synthetase. Dihydropteroic acid is then converted to dihydrofolic acid by dihydrofolate synthetase. In the final step of the pathway dihydrofolic acid is converted to tetrahydrofolic acid, the active form of folic acid, by dihydrofolate reductase. Trimethoprim prevents the enzyme dihydrofolate reductase reacting with the intermediate compound dihydrofolic acid, thereby blocking the pathway in the final step.
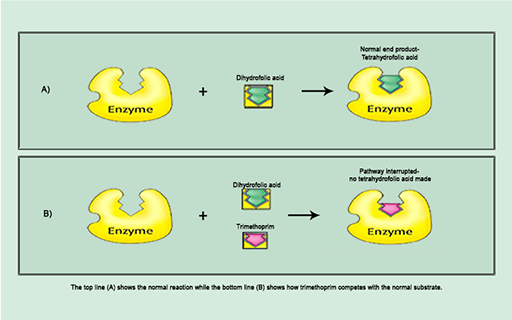
Figure 13b The underlying competitive mechanism.
Show description|Hide descriptionThis figure shows the underlying competitive mechanism by which trimethoprim inhibits dihydrofolate reductase. Part (a) shows the mechanism in the absence of trimethoprim represented as an equation in which dihydrofolic acid (in green) and dihydrofolate reductase (in yellow labelled enzyme) interact so that dihydrofolic acid binds to the active site of the dihydrofolate reductase. Part (b) shows the mechanism in the presence of trimethoprim represented as an equation. Trimethoprim (in red) competes with dihydrofolic acid (green) to bind to the enzyme (represented in yellow). When trimethoprim binds to the enzyme dihydrofolic acid is unable to bind and the pathway is interrupted.
The action of trimethoprim illustrated in Figure 13b exemplifies the specific interaction between antibiotic and bacterial target at a molecular level that disrupts a particular cellular process.