4.3 Drivers of resistance
Activity 18: Resistance to newly discovered antibiotics
Listen to part of a TED talk (between 3:49 and 5:00 in Video 8) and pay particular attention to the figure that appears onscreen at around 4:45, which summarises the development of resistance against different classes of antibiotics.
Transcript: Video 8
Briefly, it works like this. Bacteria compete against each other, for resources, for food, by manufacturing lethal compounds that they direct against each other. Other bacteria to protect themselves, evolve defences against that chemical attack.
When we first made antibiotics, we took those compounds into the lab, and made our own versions of them. And bacteria responded to our attack, the way they always had.
Here is what happened next:
Penicillin was distributed in 1943, and widespread penicillin resistance arrived by 1945.
Vancomycin arrived in 1972; vancomycin resistance in 1988.
Imipenem in 1985 and resistance to it in 1998.
Daptomycin, one of the most recent drugs, in 2003, and resistance to it just a year later in 2004.
For 70 years, we played a game of leapfrog: our drug, and then resistance. And then another drug, and then resistance again.
The more antibiotics we use, the more we select for resistance. Any
This process holds true for any newly discovered antibiotic. Evolution means that resistance will inevitably emerge, but it will do so more rapidly if we do not use antibiotics with care. Let’s look at some graphs on
Activity 19: Human antimicrobial consumption
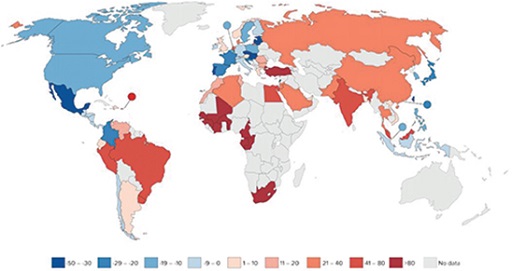
Study Figure 7 and answer the questions that follow. It shows the percentage change in antibiotic consumption per capita between 2000 and 2010. Percentage decrease is indicated in blue while percentage increase is indicated in red. Lower percentage changes are indicated by lighter colours. No data are available for the countries shown in grey.
- Is human antibiotic consumption data available for the country that you live in?
- What global trend(s) can you identify in human antibiotic consumption?
Discussion
In some countries the consumption of antibiotics by humans has decreased between 2000 and 2010, and in others it has increased. Decrease of antibiotic consumption is seen in the Americas, most countries in Europe and in eastern Asian countries. Increase in antibiotic consumption is seen in South America, Africa and Asia.
Generally, based on Figure 7, you can say that in high-income countries the antibiotic consumption by humans has decreased between 2000 and 2010, whereas it has increased in low- and middle-income countries. However, no information is provided on the use of antibiotics, such as who they were given to and whether they were used appropriately. Understanding how antibiotics are used is another important factor in understanding the drivers of AMR.
Antibiotics are also used to keep animals healthy. This includes both companion animals and food-producing animals. Use of antimicrobials in companion animals is likely to have less impact on AMR than antimicrobial use in food-producing animals. Companion animal medicine is usually based on therapeutic treatment of a small number of individual animals, whereas antimicrobial use in food-producing animals sometimes occurs at a larger scale and may include use for prophylaxis or growth promotion. In addition, treatment of food-producing animals may result in resistant bacteria entering the food chain.
The Food and Agriculture Organization (FAO) states that estimates of the total use of antimicrobials in agriculture vary considerably. This is mainly due to a lack of systems in place to collect information on use of antimicrobials in animals, although in Section 5.3 you will learn that global efforts are being made to address this data gap. Antimicrobial use in livestock and in other food-producing animals such as aquatic animals is projected to increase over the coming decades based on the increased demand for animal-sourced food products (FAO).
Activity 20: AMR in aquaculture
Watch Video 9 (up to 0:50), which looks at AMR in aquaculture:
Transcript: Video 9
Why antimicrobial resistance (AMR) in aquaculture matters for the One Health approach.
Aquaculture is one of the fastest-growing food production sectors worldwide, especially in low- and middle-income countries. But as aquaculture intensifies to meet global demand, so does the disease burden affecting aquatic animals. Antibiotics are not a long-term solution and tend to be misused without proper training and consultation, often leading to resistant bacteria: a form of AMR. AMR refers to any microbe that develops resistance to the substance designed to control it.
Antimicrobials are given to food-producing animals. Drug-resistant bacteria develop in animals; drug-resistant bacteria can spread in the environment and food, and can be transferred to people by eating food.
Resistant bacteria is a major concern to human, animal and environmental health.
Why should we be concerned about using antimicrobials in (aquatic) animals? For inspiration for your answer, look at the figure that you studied in Activity 17.
The link between use of antimicrobials in food-producing animals and AMR spread among humans requires further study, because the link isn’t as clear as we think. There is some evidence that use in animals contributes to AMR spread in people, but what isn’t clear is how much it contributes compared to use in human health. Further studies are also required to quantify the volumes of antibiotics used in companion animals compared to food-producing animals. Although we might assume there are much higher volumes of antimicrobials used in food-producing animals, there are limited data available on this point, and even less on the reasons for use (treatment, prophylaxis, growth promotion). This data gap is slowly changing as countries start to improve the ways they measure and monitor the quantities of antibiotics and how they are used in different sectors.
In conclusion, global antibiotic consumption has increased since 2000 and is predicted to continue increase in the future (WHO, 2015b). In some countries, antibiotic use in humans and animals has decreased, but the opposite has happened in others. It is important to remember that any encounter a bacterial population has with an antibiotic may select for resistant bacteria. This is irrespective of whether bacteria are innately resistant or have become resistant due to mutation or gene transfer. The antibiotics kill the non-resistant bacteria, but the resistant bacteria survive. As described above, we don’t yet fully understand the links between use in agriculture and resistance in human disease. There are proven examples of resistant bacteria in animals transferring to humans through the environment and/or the food chain; however, there is a poor understanding of the scale of these events and their impact, and it is likely that, overall, AMR in humans is related to antimicrobial use both in healthcare and in farming. All sectors, therefore, need to address the issue of misuse of antibiotics to do their part in minimising the emergence and spread of AMR.
Activity 21: Reflection on use of antimicrobials
Should we just stop using antibiotics?
Discussion
No, of course not. Antibiotics are very important to us. We just need to make sure we use them responsibly in all sectors. Also, we should look at things that we can do to limit the use of antibiotics wherever possible. There are many things we can all do, as citizens and in our professional lives, to address the problem of AMR. In the next section you will learn about this.
4.2 Spread of resistance in bacteria and the environment