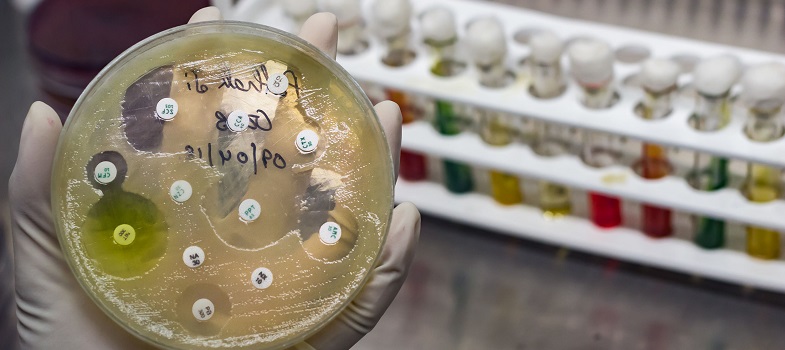
5.2 Standard operating procedures (SOPs)
Following well-written SOPs can minimise the likelihood of variables affecting the outcome of a test by including step-by-step instructions and quality procedures. You will learn about SOPs in the Quality assurance and AMR surveillance module.
Activity 9: SOPs in your workplace
If your workplace uses the disk diffusion method for AST, look at the SOP for this now. If you do not have access to an SOP in your workplace use one of these examples.
- EUCAST Disk Diffusion Test Methodology [Tip: hold Ctrl and click a link to open it in a new tab. (Hide tip)] , clinical microbiology laboratory SOP created by EUCAST (2020b)
- Standard Operating Procedures: Bacteriology – Veterinary Sector, veterinary microbiology laboratory SOP created by the
Indian Council of Medical Research (ICMR) (note that you should only refer to Chapters 4, 6 and 7 of this document for this exercise)
Find examples in your SOP of instructions or details that control the factors affecting disk diffusion tests that you learned about in Activity 7. For example, to control the depth of agar, the SOP might state the acceptable range of agar depth in culture plates and include instructions for how to prepare culture plates. Similarly, does your SOP contain guidance to cover all the factors mentioned in Activity 7?
5.1 Factors affecting disk diffusion tests