4.1 The PDCA model
The
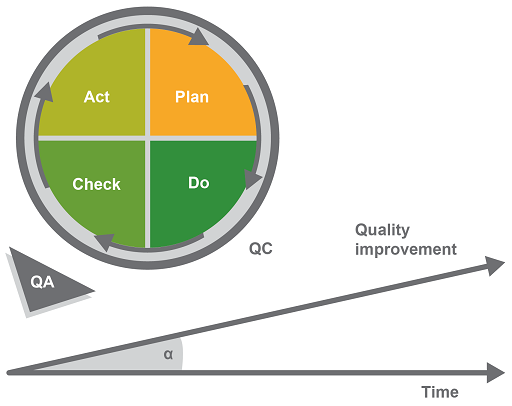
The model has four phases:
- Plan identifies opportunities for improvement and defines an action plan.
- Do corresponds to the pre-analytical, analytical and post-analytical phases of the workflow in a biomedical laboratory. (This may also be called the implementation phase.)
- Check analyses the efficacy of any new measures that have been implemented to see if the process is being improved.
- Act analyses the information from the ‘Do’ and the ‘Check’ phases, validates the approach and sets up corrective and preventive actions, thus improving the overall quality of the QMS.
Preventive and corrective actions
Audits (either internal or external) are formal processes which examine laboratory practices against established standards or regulations. The aim of any audit is to identify areas that are not performing as well as the rest of the laboratory, suggest how to improve turnaround times, and/or streamline workflows so that the laboratory is more efficient and can process more samples. Audits are an excellent method of actively looking for ways to improve a laboratory’s function.
A QMS should identify, log and investigate errors and take measures to address the root cause of a problem. Preventive action addresses problems that have not yet occurred, such as checking a piece of equipment, whereas corrective action is taken to prevent errors from reoccurring. Depending on the severity of the error, actions may range from simply checking a piece of equipment to a more formal investigation which may be necessary if the error has resulted in actual harm. The investigation and measures taken should all be clearly documented.
4 Continual improvement




