Resource 3: Lesson plan on solubility
![]() Teacher resource for planning or adapting to use with pupils
Teacher resource for planning or adapting to use with pupils
Disappearing acts
This is one way that you can talk to your pupils about the idea of solubility.
Think of a sweet like a strong mint. You put it into your mouth and enjoy the sweet, sharp, pepperminty taste. But it doesn’t last forever. It gets thinner and smaller and eventually it is all gone. Where did it go? Into the solution of sugar and saliva you have been swallowing while the sweet is in your mouth. Your liquid saliva has been gradually dissolving (the process) the solid sweet as you suck it. The result of the dissolving process is a solution of sugar and saliva.
Perhaps you could have a competition in your class to see who can hold a soluble sweet in their mouth for the longest possible time. How did the winners do it?
One little chew and the sweet breaks up into smaller pieces – you have lost! Swirl it around in your mouth and you will have lost too! But if you can hold it in the cooler air on the tip of your tongue, where there is also less saliva, you might just be a winner.
Examples like this will give your pupils some ideas about the factors that affect solubility.
Investigating solubility
1. Each group of pupils chooses a factor around solubility to investigate. Some ideas are:
- temperature of the water (try hot, warm, room temperature and icy);
- changing the size of the sugar pieces (try four lumps: one lump, one lump broken into small pieces, one lump in grains and one lump which has been ground into powder);
- stirring (try no stirs, slow stirring and fast stirring).
2. Put these questions on the chalkboard to help your pupils plan their investigations:
- What do you want to find out?
- What do you predict will happen?
- What equipment will you need?
- What will you change each time?
- What will you keep the same (to make sure the test is fair)?
- What will you actually measure?
3. Make sure each group keeps all the other factors the same (constant). Only one factor must be different for each group. For example, if they are investigating stirring, they must use the same amount of water at the same temperature and the same amount of sugar each time.
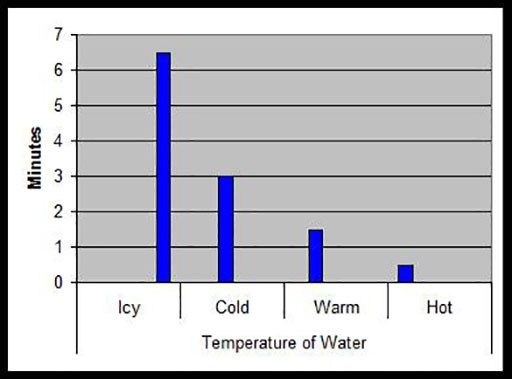
4. Ask your pupils to record their results in the form of a bar graph such as that below, which shows how many minutes it took for one spoonful of sugar to dissolve in the same volume of water at different temperatures.
Model of dissolving
This is a clearer picture of what happens when solid matter dissolves in liquid. A soluble solid comes into contact with a liquid solvent. The liquid ‘unglues’ the particles of the solid, so they can move, mix in and slip freely between the particles of the liquid.
The factors that affect the time it takes for a solid to dissolve are:
- temperature of solvent (faster in hot water);
- size of solute (faster with small grains);
- movement of solvent (stirring makes dissolving fast).
An example – dissolving salt
The teaspoon of salt is the solute. The water in the jar is the solvent. Once the salt is added, the process is called dissolving. The product of dissolving is a saline (salty) solution.
If more salt is added until no more will dissolve, we have a full or saturated solution.
Resource 2: Looking at properties of solids



