2.4 Being an atmosphere detective
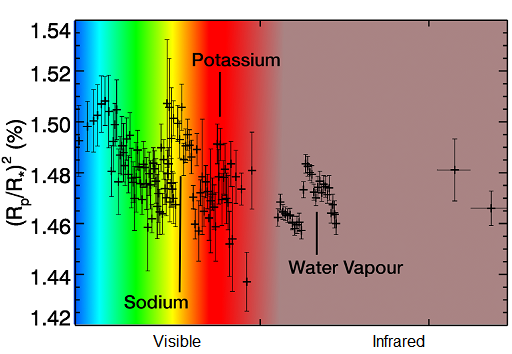
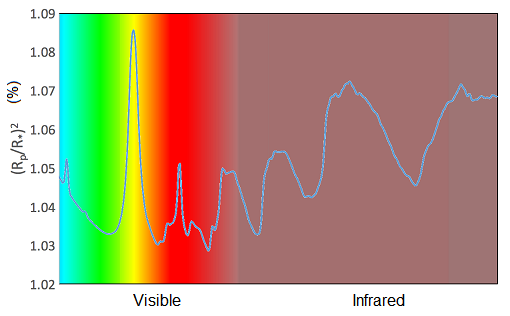
A transmission spectrum is a graph of transit depth plotted at different wavelengths. Remember that the wavelength of light corresponds to its colour. The tiny differences in transit depth appear as little wiggles in this graph. The graph in Figure 8 shows how the transit depth changes with wavelength for a planet you’ve already studied – HD 209458 b. Remember, HD 209458 b is a hot Jupiter – a big ball of hydrogen and helium gas, with traces of a few other gases mixed in. It’s nothing like the Earth, so we’re not expecting to see oxygen, and we don’t. We see evidence of water vapour – water in its gaseous form – which might not seem too strange, but we also see sodium and potassium. Sodium and potassium are both metals and they’re solid in normal conditions on Earth, but in the atmospheres of these planets they are hot enough to be gases.
Each point in the transmission spectrum of HD 209458 b is a measurement made in a particular colour of light, and the vertical bars show the level of uncertainty on each measurement – it expresses a possible range of values that measurement can occupy. The transit depth is plotted on the vertical axis, expressed as a percentage. Note that the numbers towards the top of the axis are bigger, corresponding to a deeper transit, and hence more absorption of that particular colour of light. You can see small bumps in the spectrum where sodium, potassium and water vapour absorb some of the starlight. There are gaps in the spectrum because instruments on spacecraft can only observe limited ranges of colours.
You may be wondering where the hydrogen is – after all, hydrogen is by far the most plentiful element in the Universe and is expected to be the main constituent of gas giant atmospheres, as it is for Jupiter. Although there is a lot of hydrogen, it doesn’t absorb light when in molecular form. This is the form it would take in planetary atmospheres and so it is very difficult to detect.
From these kinds of atmospheric studies astronomers have inferred that HD 209458 b and other hot Jupiters have ‘puffy’ atmospheres. This is due to the intense heating from their close parent stars (Figure 9). You may recall that in Week 5 you calculated that HD 209458 b is less dense than Jupiter, and this is probably the reason.
Activity 1 Atmosphere detective
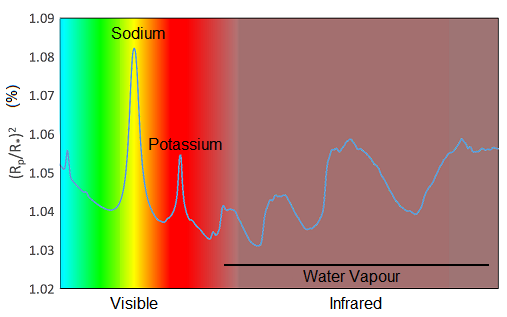
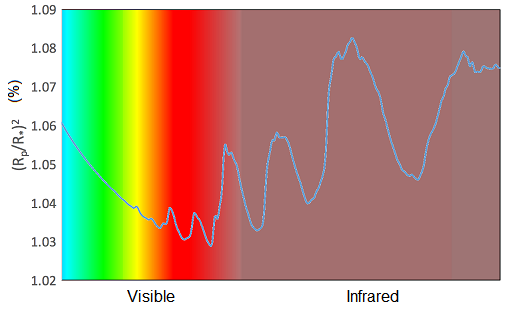
Now it’s your turn to see if you would be a good atmosphere detective. You’ll be shown four different spectra with different combinations of the gases sodium, potassium and water vapour detected. It’s your job to identify which of these gases are present in the atmosphere of planets. Take a look at the example in Figure 10 to help you decide.
This is a template showing features due to sodium, potassium and water vapour. Sodium and potassium each have one prominent feature, whereas water vapour has several broader features.
Answer
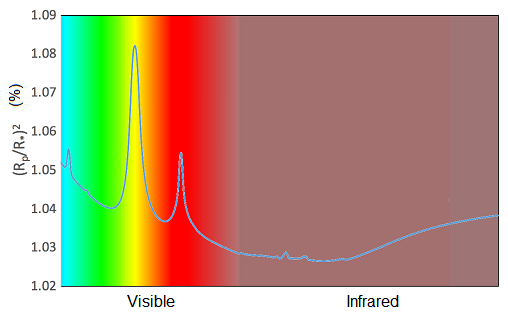
Sodium and potassium
Answer
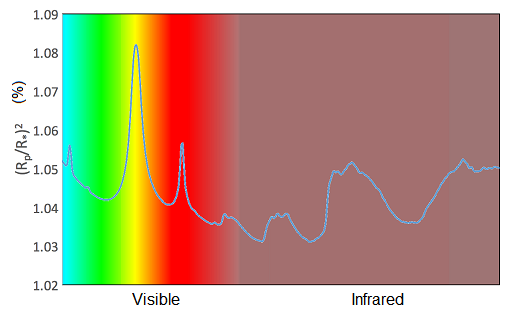
Sodium, potassium and water vapour
Answer
Sodium, potassium and water vapour