1.2 Hops
The hops used in brewing are the flowers (referred to scientifically as the strobili) of the female hop plant Humulus lupulus, a member of the hemp family (Figure 2).
Hops are used mainly as a flavouring and stability agent in beer, to which they impart bitter, zesty or citric flavours. Although primarily used in brewing, hops are also known to have various sedative, hypnotic and antibacterial properties.
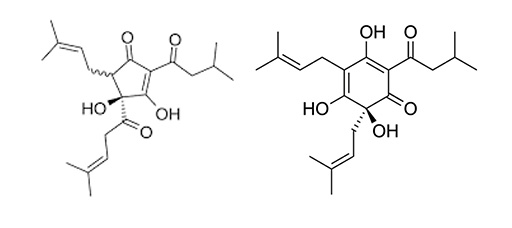
Hops contain a range of different chemical compounds that affect the flavour, the most important being the alpha and beta acids. Alpha acids such as humulone (Figure 3a) act as a mild antibacterial agent. When heated during the brewing process, it changes chemical structure into iso-humulone (Figure 3b), which imparts a bitterness into the beer.
Humulone and iso-humulone are said to be isomers of each other as each compound has the same number of constituent atoms, but a different three-dimensional arrangement of these atoms.
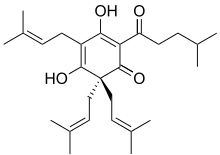
On the other hand, beta acids such as lupulone (Figure 4) are essential oils and act as aroma and flavour compounds that give the beer a distinctive taste and smell (often described as ‘hoppy’). Oils such as lupulone are volatile which means they will readily evaporate during the boil. So if a brewer wishes to produce a more ‘hoppy’ beer, great care must be taken about when the lupulone is added to the boil. If added too early, it simply evaporates and the flavour is lost.
Look carefully at the structures of both components of the hops. Do you recognise a particular functional group?
Both humulone and lupulone contain the –OH functional groups and so are further examples of types of alcohol, although their names are derived from other functional groups within the compounds.
Because different types of hop have different combinations of alpha and beta acids, hop growers must have their produce analysed by a laboratory before the hops can be sold, so the brewer knows how they will affect the beer. In order to create a beer with an optimal combination of bitterness, flavour and aroma, the brewer must take into account the ratio between the two types of acid.