What is an electric current?
Within every atom there’s a tiny, positively charged nucleus surrounded by a number of even smaller, negatively charged particles called electrons. The electrons each carry the same amount of negative charge. The charge on the nucleus is equal in size to the total charge of all the electrons in an atom, which is why atoms are electrically neutral overall.
The reason the nucleus and electrons of an atom stay together is exactly the same reason that your hair is attracted to a hairbrush – the electric force pulls them together. However, atoms can lose or gain electrons to leave themselves with a net positive or negative charge, and they are then called ions.
In an electrically conducting solid, like a metal wire, electrons can move around fairly freely. We can therefore picture such a solid as consisting of a sea of negatively charged electrons moving randomly through a fixed array of positively charged ions.
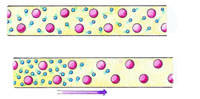
A fixed array of positively charged ions (red) surrounded by a 'gas' of negatively charged ions (blue) in an electrically conducting wire. The positive charges on the ions and the negative charges on the electrons balance, so the solid is electrically neutral.
The electrons on the left will migrate in a conducting wire under the action of electric forces, due to the change imbalance at the ends. The arrow shows the direction of movement of the electrons.
Now let’s consider what would happen if we concentrated all the electrons at one end of the wire. There would then be a large excess of positive charge at one end, and a large excess of negative charge at the other. The electrons, which are free to move, would rush back towards the positively charged end of the wire, until their negative charges once again just balanced the positive charges. This flow of negatively charged electrons appears as an electric current in the wire. In this case, the electric current would peter out very quickly, as soon as the electrons had redistributed themselves to cancel out the charge imbalance.
If we wanted to maintain a flow of electrons (an electric current) in the wire, we would need to continually suck electrons out from the ‘positive’ end and push them in at the ‘negative end’. In this way a steady state would be reached and the electrons would keep travelling along the wire, as a result of the charge imbalance. This is exactly what happens in an electric battery.
When a conducting wire is connected between the positive and negative terminals of a battery, one end of the wire becomes positively charged and the other end negatively charged. The difference in charge causes electrons to move through the wire towards the positive terminal of the battery, where they are removed from the wire. At the same time, the negative terminal supplies more electrons to the wire, so the charges don’t continually build up at the battery terminals. In other words, the battery acts as a kind of pump, pulling electrons from one end of the wire and pushing them into the other. The energy required to drive this process comes from the chemical reactions that take place inside the battery.
So batteries are just devices that convert chemical energy into electricity. To kickstart the chemical reactions in the battery, you just connect a wire between its negative and positive terminals, and a steady stream of electrons (a current) is produced as the reactions get under way. If an electrical appliance, like a light bulb, motor or radio, is placed between the terminals, the current generated will power the device. The faster the flow of electrons, the greater the current.
Because the chemical reactions will only start when the terminals are connected, an unused battery can sit on a shelf for a year and still pack plenty of power. However, a battery only contains a fixed amount of reactants, and, once these have been used up, the chemical reactions stop – the battery is dead!
a battery
THE FIRST BATTERY
The first ever battery was demonstrated in 1800 by Count Alessandro Volta. He found from experiments that different metals in contact with each other created electricity. He therefore built a stack of alternating discs of zinc, blotting paper soaked in saltwater, and silver (or copper, depending on who you believe). When he attached a wire to the top and bottom discs (which must be different metals for it to work) he measured a voltage and a current. He found that the pile could be stacked as high as he liked, and that each additional layer increased the voltage by a fixed amount, depending on the metals he used.
The current is produced because of a chemical reaction arising from the different electron-attracting capabilities of the two metals. This device became known as a 'voltaic pile' (the French word for 'battery' is 'pile'). Although they were large and bulky, voltaic piles provided the only practical source of electricity in the early 19th century.
MAKE YOUR OWN PILE
You can create your own voltaic pile using 2p and 10p coins and some paper towel the same size as the coins and soaked in lemon juice (or water that has had as much table salt dissolved in it as possible - what's called a 'saturated' solution). The paper discs need to be the same size as the 10p coins so that they don't overlap and short-circuit. Build up your pile by placing a 2p coin, a 10p coin and then a disc of wet paper towel in sequence. The lemon juice (or salt solution) is there to act as a conductor, to help the electrons travel between the different metals.
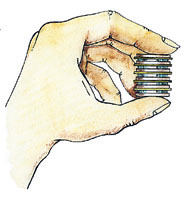
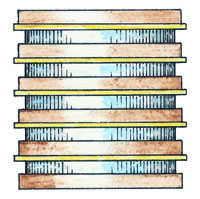
home-made voltaic pile
Repeat the 2p coin/10p coin/wet paper sequence at least ten times. If you have a multimeter, you should be able to measure the voltage. You can construct a pile using any two different metals. You'll find that different combinations of metals will produce slightly different voltages, though you won't be able to feel the difference.

Rate and Review
Rate this article
Review this article
Log into OpenLearn to leave reviews and join in the conversation.
Article reviews