2.2 Non-molecular substances
Non-molecular substances defy attempts to pick out discrete molecules from their structures.
You have already met sodium chloride (NaCl) earlier in this module – this is common table salt.
-
What chemical entities make up NaCl?
-
Sodium cations (Na+) and chloride anions (Cl-)
But how are these ions arranged to form a crystal of sodium chloride?
This is built up from tiny cubes shown in Figure 5 consisting of sodium cations (Na+) and chloride anions (Cl-)
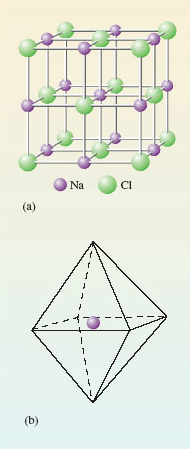
Look first at the sodium at the centre of the cube. The six chloride anions lie at the corners of a regular octahedron Figure 5(b).
The formula NaCl is an empirical formula; so it is telling you that in sodium chloride there are equal numbers of sodiums and chlorines. This condition is automatically fulfilled by joining many of the cubes shown in Figure 5(a) through their faces.
But, Figure 5 provides no evidence that NaCl is the molecular formula of sodium chloride.
In fact quite the opposite.
-
Suggest why this is the case?
-
The six chloride ions around the sodium cation all lie equal distances away – remember this is a regular octahedron. There are no grounds for singling out just one chloride ion, coupling it with a sodium cation and calling the result an NaCl molecule.
So as there is no evidence of discrete NaCl molecules in the solid; NaCl is a non-molecular compound, and the concept of a “molecular formula” is not appropriate.
