1.1 How do we find out the energy in food?
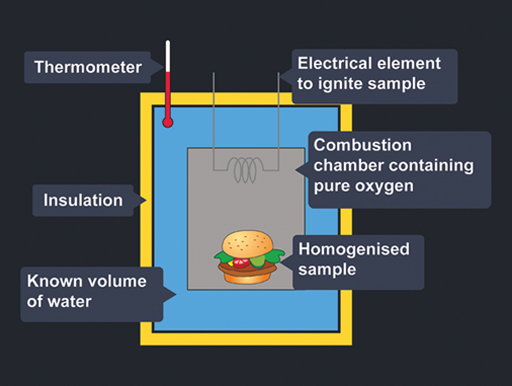
Figure 4 is a cross-section through the apparatus that is used to find the amount of energy in food. It is called a bomb calorimeter.
The ‘bomb’ refers to the thick-walled container used, which is filled with pure oxygen at a pressure of about 30 atmospheres. The food has to be homogenised, dried and weighed before being put into the chamber. You can’t use a whole burger like this!
When the power is switched on, the electrical element causes the sample to burn completely in the oxygen. The heat given off by the burning food heats up the water surrounding the chamber. The temperature rise is detected by the thermometer.
From this temperature increase, and the volume of the water, the amount of energy given off by the combustion of the food can be calculated. The number of kilocalories in the food can be calculated easily because 1 kilocalorie will raise the temperature of 1 kilogram of water by 1 °C.
However, it is not quite this simple. Combustion in a bomb calorimeter burns everything in the sample and all the energy is released. But this is not the case in the human body. Dietary fibre, for instance, is not used for energy in the body but passes through unchanged. So the value on the food label is usually about 85% of the value obtained by calorimetry.
The typical calorie content of the main food types is shown in Table 1.
| Food type | Energy (kcal/gram) |
|---|---|
| Fat | 9 |
| Protein | 4 |
| Carbohydrate | 4 |
| Alcohol | 7 |