This content is associated with The Open University's Science courses and qualifications
The principles of the TIG welding torch
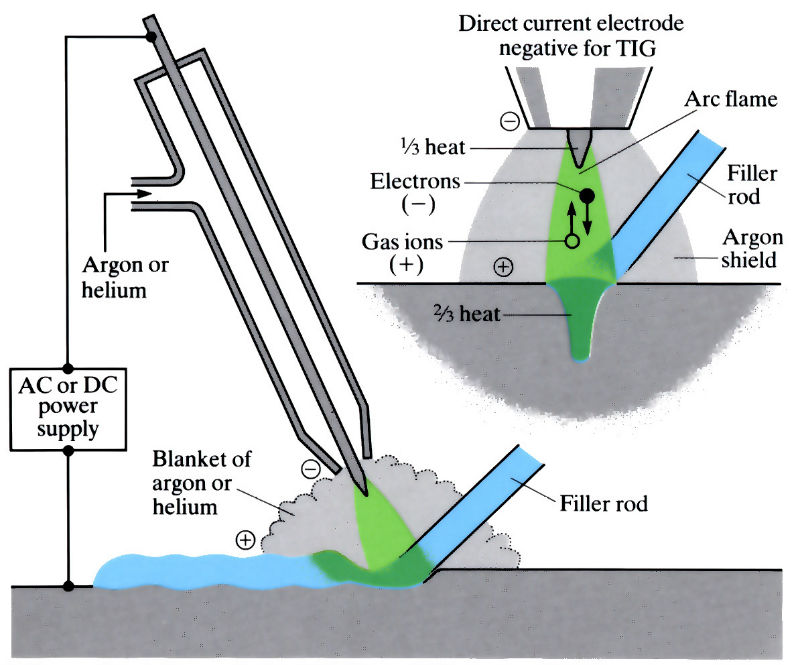
Tungsten inert gas welding (TIG) is an arc welding process, which uses a non-consumable tungsten electrode, shrouded by a protective atmosphere of inert gas, such as argon or helium. The extra weld metal can be provided by a separate filler rod if necessary. A direct current is used, with the electrode negative, to avoid overheating and undue erosion of the tungsten electrode.
The principles of a MIG welding “gun”
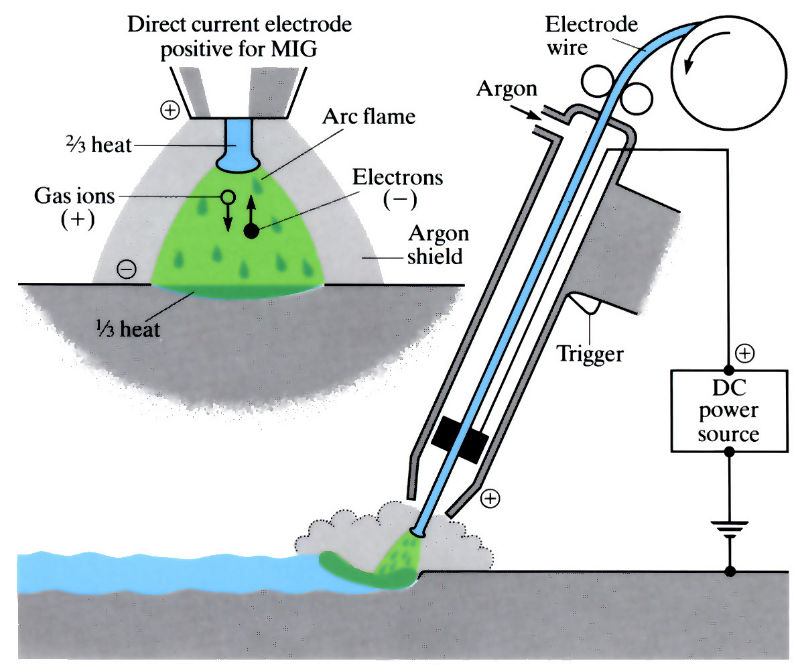
The metal inert gas (MIG) process uses a consumable electrode, which is usually in the form of a copper-coated coiled wire. Argon is used to shield the weld, and direct current with the electrode being positive to generate more heat for melting.
Manufacture:
TIG welding
- Uses a non-consumable electrode of tungsten with addition of 1% thoria (ThO2).
- Process is particularly useful in welding thin sheet, without the need to use filler metal.
- High purity argon gas produces welds free of oxidation, which enables reactive metals such as titanium and zirconium to be successfully welded.
- No slag formation eliminates post-cleaning operations.
- Minimal weld splatter.
- Slower than MMA or MIG processes.
- Good control of welding current, arc length and filler metal additions.
- Lends itself to mechanisation.
- Power supply up to 300A a.c. or d.c.
MIG welding
- Process can be semi-automatic or automatic.
- Feed wire diameters range from 0.75 to 2.25 mm.
- Wires are usually copper-coated to improve conductivity.
- Argon/helium mixtures can be used for shielding.
- Produces high quality welds at high speeds, and no flux to remove (deposition rates 1.25–7.5 kg h-1).
- Power supply 60–500 A, 16–40 V d.c.
Metal active gas (MAG) and CO2 welding
- Argon or helium is replaced in the MIG process by carbon dioxide (with or without additions) at lower cost.
- CO2 is used mainly for the welding of steel.
- Addition of up to 10% oxygen to the CO2 base gives the following advantages: provides smoother transfer of weld metal, increases the fluidity of the weld pool and increases the wettability of the weld metal.
Materials:
TIG welding
- d.c. welding used for carbon and alloy steels, heat resistant and stainless steels, copper and its alloys, nickel and its alloys.
- a.c. welding is required to TIG weld aluminium, magnesium and aluminium-bronze alloys to break down the tenacious surface oxides on the metal surface.
- Use of high purity argon enables reactive metals such as titanium and zirconium to be welded, with argon shrouds and d.c. current.
- Thin walled (1.6 mm and less) stainless steel tubing can be TIG welded by rotating the welding head and fixing the tube. This is called orbital pipe welding.
MIG welding
- MIG process is suitable for welding aluminium, magnesium alloys, plain and low-alloy steels, stainless and heat-resistant steels, and copper and bronze.
- Variations are in the filler wire composition, current and voltage and shielding gas.
MAG and CO2 welding
- CO2 welding is mainly used for welding mild steel and low-alloy steels (cheaper than argon).
- CO2 is really effective as a shield gas if the electrode wire contains up to 1.8% manganese, 0.5% silicon, 0.15% titanium and 0.15% zirconium, which act as deoxidising agents.
- Stainless steels are MAG welded with argon +1% oxygen.
Design:
TIG welding
- Can successfully weld thin-gauge materials with minimal distortion (< 0.5 mm thick).
- Aluminium alloys with plate thicknesses of 2–6.4 mm can be welded as flat butt joints. Plate with thicknesses of 5–9.5 mm welded with single V-butt joints.
- Thin section stainless steel tube can be TIG welded by orbital pipe welding.
MIG welding
- Plate thicknesses of 6–25 mm can be flat-butt welded in aluminium with root faces of 1.6–4.8 mm.
- Productivity is higher than TIG welding.
- Used in general engineering construction.
MAG and CO2 welding
- Automatic welding by MAG or CO2 processes produces consistent high quality welds in mild steel and low-alloy steels.
- Higher speeds of welding than by TIG or MIG.
This article is a part of Manupedia, a collection of information about some of the processes used to convert materials into useful objects.



Rate and Review
Rate this article
Review this article
Log into OpenLearn to leave reviews and join in the conversation.
Article reviews