1. Monomer or low molecular weight polymer, or mixture of both, mixed with initiator (catalyst), inhibitors, fillers, etc.
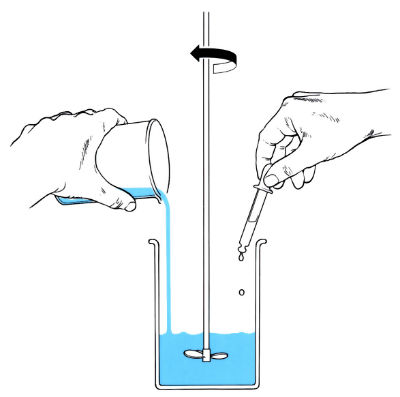
2. Mixture poured into open mould.
3. Mould heated or cooled during chemical reaction.
4. Moulding removed.

Manufacture:
- Minimum capital expenditure.
- Moulds can be epoxy, plaster, wood, sheet metal, or even rubber.
- Difficult to automate, but ideal for labour-intensive production of chess pieces, garden ornaments, etc.
- Extensively used for the production of very high molecular weight sheet needed for the thermoforming of, for example, aircraft windows.
- Process forms the basis for encapsulation of electronic components, hand lay-up of composite structures and adhesive bonding.
Materials:
- The starting materials must possess low viscosity for pouring.
- Process can be used for thermosetting polymers, unsaturated polyesters, epoxies, etc., thermoplastics, especially vinyl polymers such as polymethyl methacrylate, or rubbers, principally room temperature vulcanising (RTV) silicones and urethanes.
- Monomer, polymer or a mixture of the two (which may be the same or different) can be used. Initiators will depend on the polymer, but fillers and pigments may be added as long as the viscosity remains low.
- Reaction may be spontaneous or initiated by temperature. Exothermic reactions are common, and inhibitors are often included to moderate the temperature rise.
Design:
- Orientation of final polymer totally random because of the low shear applied to the mixture and the low molecular weight of the starting material.
- Possible exotherm limits section thickness to avoid degradation at centre.
- Air entrapment is a problem and, after pouring, mixtures are often subjected to vacuum to eliminate bubbles.
- Re-entrant angles are produced by flexible moulds, or by flexible mouldings.
See Also: Melt casting, Slip-casting and Gravity die casting.
This article is a part of Manupedia, a collection of information about some of the processes used to convert materials into useful objects.
Rate and Review
Rate this article
Review this article
Log into OpenLearn to leave reviews and join in the conversation.
Article reviews