2.1 The monoaminergic hypothesis for mood disorders: re-examined
The BPS-E model offers a more holistic understanding of mental health science by giving importance to all relevant domains of knowledge. This does not mean that we should not focus on a given domain and explore this in more depth. Development of greater understanding requires us to critically examine defined aspects within a given domain, as well as their relationship with other elements.
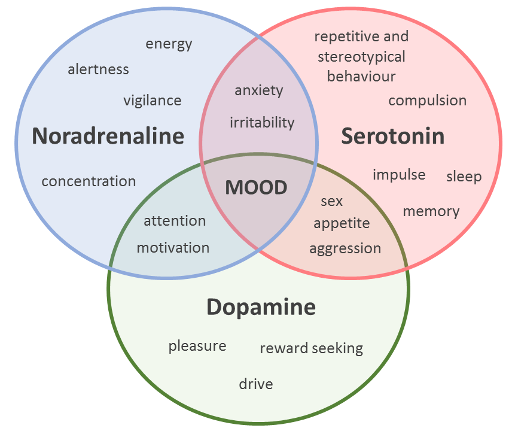
The ‘monoaminergic hypothesis’ is one such component, considered to be central to a biological understanding of mood disorders (which include both anxiety and depression). The hypothesis proposes that the underlying neurobiological basis of anxiety and depression rests on the depletion of levels of key monoamine neurotransmitters: serotonin, noradrenaline (termed ‘norepinephrine’ in the US) and/or dopamine within the brain. The hypothesis is supported by the mechanism of action of antidepressant drugs which have been shown to elevate the levels of these neurotransmitters in the brain, and are known to be effective in alleviating the symptoms of anxiety and depression.
Selective serotonin reuptake inhibitors, or SSRIs, in particular, which increase serotonin levels (by preventing the reuptake of this neurotransmitter from synapses) can be effective for both depressive disorders and anxiety. However, of the estimated 350 million people worldwide who experience depression, between a third to a half do not improve with ‘standard’ antidepressant treatment.
Similarly, 'at least a third of patients with anxiety disorders do not adequately respond to available pharmacological treatment' (Maron and Nutt, 2015). The occurrence of such ‘treatment-resistant’ (or ‘treatment refractory’) cases, casts doubt on the primary (or sole) dysfunction of the monoaminergic system in anxiety and depression. Consistent with this view, the gamma-aminobutyric-acid (GABA) system, a target for tricyclic antidepressant medications, has long been recognised as relevant to understanding mood disorders. More recently, attention has shifted to an imbalance in glutamatergic neurotransmission with the finding that ketamine (an analgesic/anaesthetic drug) may also act as a novel fast-acting antidepressant (Kirby, 2015; Malhi et al., 2016). Considering the overlapping and inter-related functions governed by these neurotransmitter systems, it is perhaps not surprising that the picture that emerges here is complex. We have provided you with a glimpse of this in Figure 2.