1.2.3 The physiological and behavioural levels
Anticipation of winter
If organisms are to survive winter, they must be well prepared for cold weather and sometimes for reduced supply of food. Physiological changes such as shedding leaves and building up fat reserves, and behaviours such as hoarding food, must be completed before winter begins. In order to be prepared, organisms need to anticipate the onset of winter, which is done in two ways, each of which has its own associated physiological mechanisms. First, they could respond to environmental cues that predict that winter is imminent, e.g. lower temperatures and shorter day lengths.
Question 1
Which of these cues is the more reliable?
Answer
Changes in day length, which follow an identical pattern every year. We know from personal experience that temperature is very variable and that cold weather comes earlier in some years than others.
Secondly, animals could have an internal clock which tells them what time of year it is, just as a calendar tells us the date.
Photoperiodism
A great deal of research has been carried out into the way that organisms respond to changes in day length, i.e. the relative durations of light and darkness in a 24-hour period. This relationship is expressed as the ratio of the number of hours of light (L) to the number of hours of darkness (D), i.e. L : D. Photoperiodism is defined as the responses of organisms to changes in the L : D ratio. For example, flowering plants can be divided into three categories on the basis of the effect of photoperiodism on the onset of flowering:
Short-day plants flower in early spring (e.g. primrose, Primula vulgaris) or in the autumn (e.g. Chrysanthemum spp.), when L is small relative to D.
Long-day plants flower in the summer (e.g. potato, Solanum tuberosum, or lettuce, Lactuca sativa), when L is large relative to D.
Day-neutral plants are not affected, in terms of flowering, by changes in the L : D ratio (e.g. groundsel, Senecio vulgaris).
Horticulturalists have long been familiar with these effects and exploit them to get plants to flower at times that suit them. If you go to the Chelsea Flower Show, which takes place in May, you see spring, summer and autumn plants, raised under artificial conditions, all in bloom at the same time.
Question 2
How can chrysanthemums be made to flower later in the year than normal?
Answer
By keeping them under high L : D conditions during the spring, using artificial lights, and transferring them to low L : D conditions just before they are required to bloom.
Simply observing that primroses, for example, flower in early spring is consistent with the hypothesis that they flower in response to short light periods, but it does not preclude alternative hypotheses, such as that they flower in response to increased temperature. However, primroses kept indoors under consistently long light periods and a variety of temperatures do not flower, confirming that onset of flowering is indeed a response to short light periods. Photoperiodism can only be revealed by means of experimental manipulations in which plants are kept under different L : D regimes, and in which other environmental cues to which they might respond are held constant. Variations on this kind of experiment tell us other things about photoperiodism. For example, some plants flower in response to a single exposure to an appropriate L : D ratio. Others flower only if an appropriate L : D ratio is sustained for several days. A pigment, called phytochrome, which switches between 'dark' and 'light' forms, is involved in sensing the length of the light and dark periods.
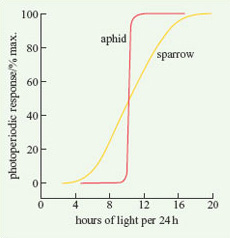
For a specific physiological response, there is typically a particular L : D ratio at which the response starts to occur. The critical photoperiod is defined as that L : D ratio at which 50% of the population being studied switches from one state to another. There is considerable variation in the preciseness of a particular organism's response to a critical photoperiod. Figure 1 shows two examples of critical photoperiods for animals. Whereas all aphids switch from sexual to asexual reproduction at an L : D ratio of 10 : 14, male sparrows start to show testicular development at an L : D ratio of about 8 : 16, but all do not mature unless the ratio is about 16 : 8.
In some animals, the physiological basis of this effect is known. The hormone melatonin is secreted by the brain during the dark period, with the result that in winter blood levels of melatonin are much higher.
Circannual Clocks
The physiology and behaviour of microbes, plants and animals show cyclical changes on a diversity of time-scales. Most familiar to humans are circadian rhythms, which determine patterns of sleep and wakefulness and of changes in body temperature over a 24-hour period. There is also evidence that certain annual rhythms are controlled by internal clock mechanisms, called circannual clocks (circannual means 'about a year').
Whatever the time-scale over which they operate, the existence of endogenous clocks can only be determined by experimental investigation. Simply observing that an animal or a plant shows a daily or an annual cycle of activity or physiology does not preclude the possibility that it is responding to rhythmic changes in the environment. The crucial experiment is called a free-running experiment, in which organisms are kept under conditions in which they are unable to detect normal cycles in external features of their environment. For example, captive alpine marmots (Marmota marmota, a member of the squirrel family) have been kept for long periods under constant temperatures and constant L : D ratios. Despite having no known cues that winter was approaching, they showed a 100% increase in food intake in the autumn, just as they do in nature, in preparation for hibernation. Kenagy (1981) found that chipmunks (Eutamias minimus and E. amoenus, also in the squirrel family) kept under combinations of three photoperiods (L : D ratios 8 : 16, 12 : 12 and 16 : 8) and two temperatures (5 and 23°C) maintained normal cyclical patterns of testis growth, body mass, water consumption, locomotion and winter torpor.
Linkage of animal reproductive cycles to winter
The reproductive success of organisms is crucially determined by the time of year when they breed. Many birds living in Britain breed in the spring, with the result that they are able to feed their young at that time of year when there is most suitable food available. For many kinds of animal, all parts of the reproductive process, from mating to birth, follow the end of the winter. A complication for some larger mammals, however, is that there is a long gestation period (the interval between conception and birth, nine months in humans). Along gestation period and giving birth early in the spring are not easily reconciled with being inactive in winter. Figure 2 shows how the reproductive cycles of four mammals native to Britain are related to the winter.

The hedgehog (Erinaceus europaeus) has a short reproductive cycle and so can complete the entire process in the spring, mating soon after emerging from hibernation. The red deer (Cervus elaphus), with a long gestation period, mates in the autumn (the rut) and gives birth in spring, remaining active through the winter. The other two species shown in Figure 2 have gestation periods that are too long for the entire reproductive cycle to be completed in the spring, but too short to occupy the whole winter. Badgers (Meles meles) mate in the autumn and the eggs are fertilised immediately. Implantation of the zygote into the wall of the uterus is, however, delayed for several months, during which the female spends short cold spells in a state of torpor in an underground den. The noctule bat (Nyctalus noctula) mates in the autumn but the eggs are not fertilised. Instead, females store sperm in their reproductive tract until late winter, when fertilisation occurs and gestation begins. Throughout this time, they are hibernating.
These different reproductive patterns are controlled by environmental cues in much the same way as the flowering of plants. Red deer, badgers and bats are called short-day breeders; the development of their gonads and their sexual behaviour is stimulated by the lengthening dark period characteristic of autumn. Hedgehogs and many other small mammals are long-day breeders; their gonad development and sexual behaviour are triggered by a decrease in the dark period.
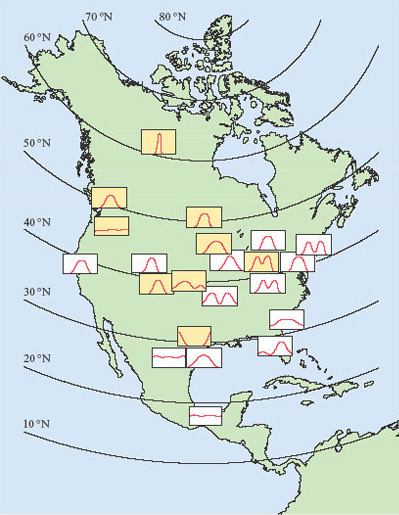
Figure 2 illustrates how diverse are the reproductive cycles of a single group of animals, the mammals. Deer, badgers and hedgehogs are very different animals, however, and before we leave this brief account of seasonal breeding, it is important to emphasise that there is enormous diversity, even among closely related species. Figure 3 shows the breeding seasons of members of a single genus, Peromyscus, in North America. Peromyscus is a genus of small American rodents that includes a variety of mice, such as the deermouse, white-footed mouse and cactus mouse.
Question 3
How does the breeding season in Peromyscus species change with latitude?
Answer
The breeding season of Peromyscus is limited to a single, three-month period in northerly latitudes, but is continuous through the year in Mexico. In between, there is considerable variation in the duration of the breeding season; in some regions at intermediate latitudes, there are two peaks in breeding activity.
Winter fat reserves and reproduction
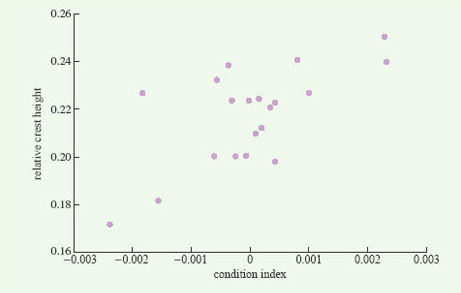
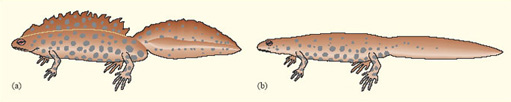
For some animals in which breeding begins as soon as the winter has finished, energy reserves accumulated in the previous autumn may be important, not only for winter survival but also for reproductive success in the following spring. In newts, for example, as in other temperate amphibians, nutrient reserves are built up in the late summer and autumn, in both the liver and the abdominal fat stores. These reserves are only partly used up in the winter, when newts are largely torpid underground, and the remainder plays a key role in reproduction. Female newts use their fat reserves to produce yolk for their eggs; males use theirs to develop a large dorsal crest, which is crucial in mating, females responding positively only to males with large crests (Figure 4). A population of great crested newts (Triturus cristatus) on the Open University campus has been studied in detail by John Baker. He weighed and measured males as they migrated towards their breeding pond in early spring and, for each male, he calculated a 'condition index'. Males in good condition (i.e. with a condition index value greater than zero) were heavy for their length, because of their larger fat reserves. Later in the year, Baker recaptured the same individuals, now in the pond, and measured their dorsal crest, which develops after newts have entered the water. He found a positive correlation between their crest height and their condition index measured a few weeks earlier (Figure 5). Thus, a male newt's attractiveness to females, and hence his reproductive success, is partly determined by the amount of fat that he has left over after hibernating during the winter.