1.3.2 Birds and mammals
Birds and mammals are endotherms, meaning that they produce and retain a lot of heat within their own tissues, rather than absorb heat from their environment, as ectotherms, such as insects and reptiles, do. The terms endotherm and endothermy are now often used in preference to homeotherm and homeothermy, which refer to the ability of birds and mammals to maintain a more or less constant body temperature. Some endotherms, as you will see later, do not maintain a high body temperature at all times, and some ectotherms, such as larger reptiles, maintain a constant body temperature for long periods, even though the temperature of their environment changes.
Whilst endothermy allows some birds and mammals to remain active during winter, it also places formidable demands on those animals. Reduced environmental temperatures increase the amount of heat that they need to generate internally to maintain a constant temperature, at a time when the amount of food available to them is much reduced. For birds and many mammals, shorter day length in winter reduces the time available for finding food. It has been estimated that a small bird, such as a great tit, which feeds on seeds and small insects, has to find food items at an average rate of one every ten seconds during daylight hours through the winter. The extreme challenge posed to birds by the shortage of food in winter is illustrated by the impact of bird-tables in urban areas, which supplement their natural diet. Studies by the RSPB (Royal Society for the Protection of Birds) suggest that, in urban areas of Britain, the provision of food at bird-tables significantly increases the winter survival rate of several garden bird species.
Maintaining body heat
Mammals that remain active in winter maintain a core body temperature of around 37-38°C; that of most birds is a little higher. In temperate habitats, thermal constancy is achieved despite the temperature of the environment varying between −20 and +20°C. For animals in polar regions, the problem is even more severe, their environment varying between −60 and +20°C. There are three principal ways in which endotherms can respond adaptively to cold conditions:
They can raise their metabolic rate so as to produce more heat to offset the increased heat loss.
They can improve external insulation between their body and the external environment, so as to reduce the rate of heat loss.
They can alter the pattern of blood circulation around their body so as to minimise the extent to which warm blood comes close to the skin (i.e. another form of insulation).
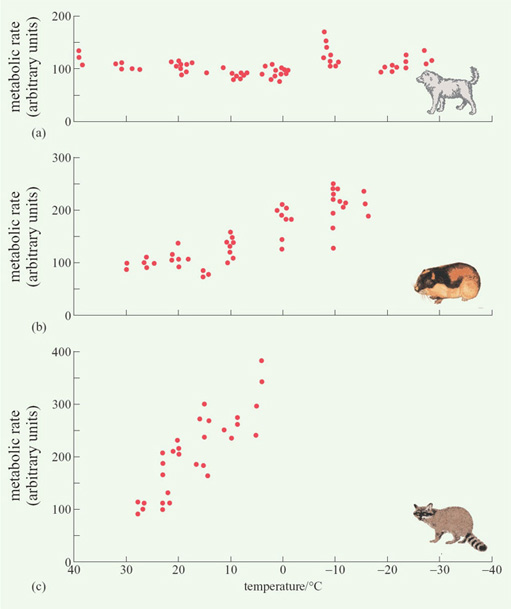
Considering the first of these processes, Figure 6 shows data from experiments in which three mammalian species, two from the Arctic and one from the tropics, were exposed to declining temperatures.
Question 4
In what way does the response to reduced temperature of (a) the Eskimo dog and (b) the arctic lemming differ from that of the tropical raccoon?
Answer
(a) The metabolic rate of the Eskimo dog shows no clear pattern of change (except possibly increasing at very low temperatures), whereas that of the raccoon increases almost linearly as the temperature decreases from 28 to 5°C. (b) The metabolic rate of the arctic lemming begins increasing at about 15°C, but more gradually than that of the tropical raccoon.
Mammals and birds living at high latitudes generally rely on processes 2 and 3, rather than 1, though, as we have seen from Figure 6(b), there are arctic species, such as the lemming, in which process 1 does play a major role.
Maintaining a high metabolic rate through a long winter requires that mammals and birds either maintain a high rate of food intake, or carry substantial energy reserves within their body, or show some combination of the two. They must also reduce heat loss during the winter. Many species maintain larger energy reserves, in the form of adipose tissue. The adipose tissue beneath the skin (subcutaneous fat) is also widely assumed to act as a thermal insulation layer, but this theory is erroneous. Adipose tissue is not much better an insulator than muscle. Only for marine mammals, such as seals, in which the adipose tissue forms a thick layer of blubber, is its role in thermal insulation significant. For birds and mammals, insulation is provided by feathers and fur respectively, which trap a layer of air next to the skin. Static air is a very poor conductor of heat, so that air trapped in plumage or fur reduces heat flow between an animal's skin and the outside. Bird plumage is a remarkably effective insulator; it represents only 5-7% of a bird's mass and trapped air makes up 95% of its total volume. The texture and often the colour of birds' plumage and mammals' fur changes with the onset of winter, in comparison to the summer. This change is effected during moult, a seasonal process in which a bird changes all or many of its feathers and a mammal replaces its summer coat with a thicker winter coat, and the converse in spring.
In birds, the plumage has two major components: contour feathers, which form the exterior surface; and down, which lies beneath the contour feathers. Most feathers consist, in varying proportions, of pennaceous (fan-like) and downy components (see Figure 7). The large feathers in the wings and tail are wholly pennaceous; many other small feathers close to the skin are pure down. In winter, the number of down feathers is markedly increased. Only in a few species has the total number of a bird's feathers been counted; hummingbirds have about 940, whereas a swan has 25 000. In the house sparrow (Passer domesticus) there are 11.5% more feathers in winter than in summer. Each feather has an individual muscle which enables it to be lifted away from the body. This capacity enables birds to regulate heat loss by altering the thickness of the air layer trapped among the feathers.

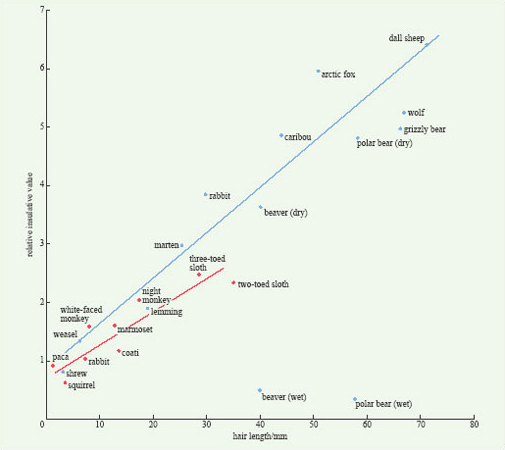
In mammals, fur contains fine hair close to the skin, underneath thicker and larger surface hairs. The insulative effect of hair is determined primarily by its length; the longer the hairs, the better is the coat as an insulator (Figure 8). Comparison of the insulative value of the pelts of arctic and tropical mammals with hair of similar length reveals that the pelts of arctic species are only slightly better insulators (Figure 8). The difference arises because arctic species have more hairs per unit area of skin. Figure 8 also shows how the insulative value of the coats of two species, the beaver and the polar bear, is completely eliminated when it becomes thoroughly wet.
In many species, the winter plumage or coat is very different in colour and pattern from the summer one. This seasonal change has nothing to do with temperature control, but is usually related to camouflage. The winter plumage of many birds, especially males, is much less brightly coloured than the summer plumage. In many polar mammals, such as the arctic fox (Alopex lagopus), the winter coat is white, making the animal well camouflaged in snow.
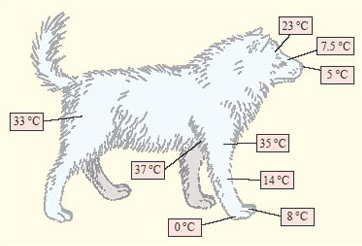
The third response to cold, varying blood flow to different parts of the body, is well illustrated by the Eskimo dog (Figure 9). At a temperature of −30°C the Eskimo dog maintains a core temperature of around +38°C, but adjustments in blood flow mean that the temperature of some parts of the body, notably the extremities such as the feet and the nose, is allowed to fall.
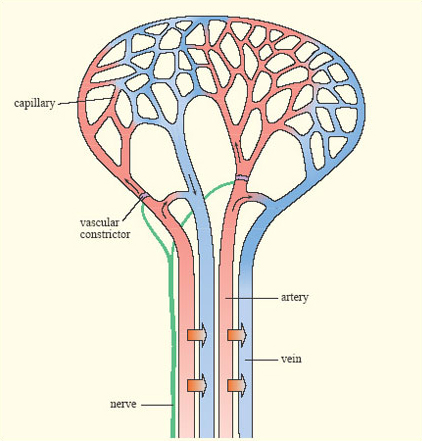
In many endotherms, cooling of the extremities is achieved by a heat exchange mechanism called a rete mirabile (pronounced 'reeta mirah-bilay' and meaning 'wonderful net', Figure 10). Warm blood passing in arteries towards the skin runs close to colder blood passing back in veins towards the body core. Thus warm blood passing outwards gives up much of its heat before it reaches the skin. This effect is enhanced by constriction of blood capillaries near the skin when it is cold.
Very small mammals and birds that remain active in winter face a problem. Because their surface area is relatively large in relation to their mass, they lose heat in cold conditions faster per unit mass than larger animals and the amount of extra fur or feathers they would need to insulate themselves would make them immobile. The dwarf hamsters (Phodopus spp.) of Siberia and Mongolia have adapted to this problem in some interesting ways. They remain active in winter but run around, feeding on seeds and vegetation, under the deep snow, where they are not exposed to the wind and the extremes of cold. Whereas larger animals typically build up their adipose tissue and put on weight as winter approaches, dwarf hamsters get markedly lighter and leaner. Males lose about 50% and females 30% of their body mass and adipose tissue falls from 35 to 5% of the total body mass.
Question 5
How might a lower body mass enable a dwarf hamster to better survive the winter?
Answer
Having a smaller body reduces an animal's energy requirement for locomotion.
Finally, consider the unfortunate pig. Pigs are large mammals and, as the result of selective breeding during domestication, they have so little hair that it provides no insulation against the cold. Pigs have the usual depots of subcutaneous fat found in other comparable mammals, but pig fat is no thicker than that of other mammals of similar size; indeed, domestic pigs have been selectively bred to be lean. The subcutaneous fat of pigs contributes little to their ability to survive cold weather. In cold conditions, the skin temperature of a pig falls much more than that of a human, due to restriction of the blood supply to the subcutaneous fat, so the poor hairless animal may feel cold in winter.
Storing food in winter
As described above, some animals solve the problem of remaining active in winter, when food is in short supply, by enlarging internal energy reserves in the adipose tissue. A problem with this strategy is that it increases body mass, which increases the energy cost of locomotion and, in extreme cases, may reduce mobility. An alternative strategy is to make external stores of food during the summer, when food is abundant, and to draw on them during the winter. Such behaviour, shown by a variety of birds and mammals, is called hoarding or caching (pronounced 'cashing').
There are two general kinds of hoarding behaviour. Larder-hoarding involves building up one or a small number of large food stores. Scatter-hoarding consists of hiding many individual food items in separate places over a wide area. An essential feature of a food cache, from the perspective of the animal that made it, is that it must not be exploited by other individuals of the same or different species. Typically, animals prevent theft by active defence in the case of larders, or by concealment if their caches are scattered. Scatter-hoarders include crows, nuthatches, tits, jays, squirrels and foxes; larder-hoarders include many small mammals, some birds, and honeybees.
Pikas (genus Ochotona) are herbivorous, guinea pig-size animals, belonging to the same mammalian order (Lagomorpha) as rabbits and hares, which live in high altitude and desert habitats of Asia and North America. They do not hibernate in winter but rely on cached food to supplement what they can find to eat beneath the snow. In late summer they gather vegetation, storing it as a large haystack in a crevice beneath a rock, which they defend against their neighbours. Laboratory experiments on a variety of mammals reveal that hoarding behaviour is triggered by cooler temperatures and shorter days which are characteristic of autumn.
Among birds, an example of a larder-hoarder is the acorn woodpecker (Melanerpes formicivorus) of western North America. During the summer, acorn woodpeckers collect acorns and store them in the branches of dead trees, drilling a hole for each acorn (Figure 11). Where oak trees are abundant, acorn woodpeckers form very stable social groups, communally defending their larders, which provide group members with food during the winter.
The time for which food is stored varies from one species to another and depends, in part, on its perishability. Coal tits (Parus ater) store insects, having removed the head and the gut, in crevices in tree bark for a few hours. They also store pellets, each one consisting of 20-50 aphids compressed together. Owls store mice for a matter of days and jays and crows store seeds for a year. Ravens store meat for several weeks during the winter, burying it under snow, which keeps it frozen and so delays decay.
The energy that animals invest in food hoarding, and the importance of their cache for their winter survival is illustrated by the following observations:
American biologist Bernd Heinrich observed an individual jay cache 127 food items in a single day.
Individual Clark's nutcrackers (Nucifraga columbina), an American bird, collect up to 95 pine seeds at a time and hide them in locations as much as 22 km from their nest.
Clark's nutcrackers breed in late winter, when there is no food available, and are totally reliant on cached food to rear their young.
Scatter-hoarding presents animals with a problem: if they hide their caches from competitors, how do they find them themselves? Leaving clues, such as scent marks, would lead rivals to the hidden food, and so they must rely on memory. Thick-billed nutcrackers (Nucifraga caryocatactes) store 15-20 hazelnuts in each cache and are able to find them even after thick snow has covered them. Detailed observations of birds in winter suggest that some species are able to memorise the exact location of several thousand stored food items over several months. David Macdonald of Oxford University studied caching in the red fox (Vulpes vulpes) by rearing a pet vixen which he took for walks in the countryside on a long lead. She frequently cached food items that he gave her, burying them in the ground. Over the course of several weeks, she subsequently found 96% of them and only rarely did she dig in places where she had not hidden food.
How do we know that animals rely on what appear to be extraordinary feats of memory to find their food caches? An alternative explanation is that they leave some kind of marker that neither their competitors nor humans can detect. Such an issue can only be resolved by carrying out experiments. Caching behaviour has been studied in American chickadees (Parus spp.) and European marsh tits (P. palustris). Captive-bred birds were trained to feed and hide food in an aviary containing artificial trees that had holes drilled in them, covered with Velcro lids. Opening these lids was very similar to the birds' natural behaviour of stripping bark off trees to find food. In the autumn, the birds spontaneously started to cache seeds in these holes. In a typical experiment, an individual bird was given 15 seeds to hide among a total of 72 open holes. When the bird had left the aviary, the seeds were removed and all the holes were covered up. Twenty four hours later, the bird was allowed back into the aviary. With very few errors, it visited the 15 holes where it had stored seeds the previous day, ignoring the others.
Question 6
Why did the experimenters remove the seeds before the bird was allowed back into the aviary?
Answer
To eliminate any possibility that the bird was using any cues coming directly from the seeds, such as odour.
Scatter-hoarding requires a remarkable memory for the location of a large number of items, a process called spatial memory. Anatomical studies of the brains of birds have revealed that the part of the brain that is involved in spatial memory, called the hippocampus, is relatively larger in species for which scatter-hoarding is particularly important. For example, the hippocampus of the marsh tit (Parus palustris) is significantly larger than that of its close relative, the blue tit (P. caeruleus), which is not a scatter-hoarder.
Changes in social behaviour
For some animals living in temperate habitats, the onset of winter also brings about a marked change in their social behaviour. Most noticeably, many birds, having spent the spring and summer as breeding pairs, being highly aggressive towards all members of the same species, other than their mate, become gregarious and form large, cohesive flocks in which individuals behave cooperatively in a number of ways. As with other biological characters, sociality confers both benefits and costs on individuals.
A major benefit of flocking is that it reduces an individual's risk of being killed by a predator. There are three main reasons for this protection. Firstly, flocks detect predators more effectively than single birds. Secondly, predators become confused when attacking prey gathered closely together. Thirdly, members of flocks may defend themselves effectively against a predator even though, individually, they are not strong enough to do so.
Being in a social group increases feeding efficiency in other ways. Firstly, individuals gain information about the whereabouts of food by observing their fellow flock-members. Secondly, animals in groups can often disturb hidden animal prey more effectively than single individuals. Thirdly, predators in groups can hunt and kill prey that are too large for them to cope with on their own. Studies of captive great tits (Parus major), using an aviary similar to that described earlier, showed that individual tits looking for hidden food found it more quickly when released into the aviary in a small flock. Individuals observed one another and avoided places where other birds had not found food, concentrating their search instead on places similar to those where other birds had located food.
For some animals, joining a social group in winter can reduce heat loss, especially on cold nights. For example, pallid bats (Antrozous pallidus) expend less energy during roosting when they roost huddled together than they do if they roost alone.
There are two major costs to becoming gregarious in winter. First, close association between individuals increases infection rates by parasites and pathogens. For example, in colonies of American prairie dogs (genus Cynomys), there is a positive correlation between the number of animals in a colony and the abundance of external parasites. Secondly, living in a group brings animals closer together, increasing the likelihood that they come into competition over food and other resources. This cost is borne particularly by smaller and weaker individuals, which generally lose in competitive interactions with other group members.
