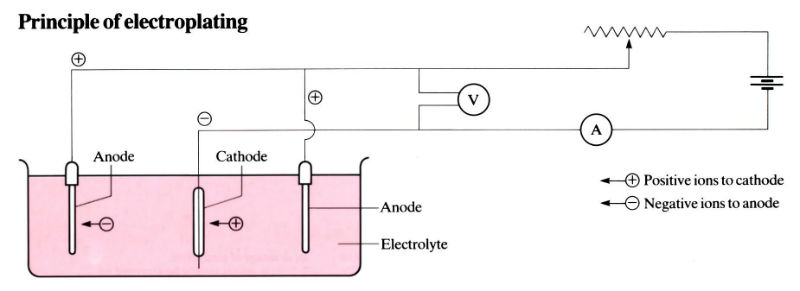
The articles to be electroplated are immersed in an electrolyte containing dissolved salts of the metal to be deposited, and they are made the cathode to a DC supply. To complete the electrical circuit, anodes are also immersed in the electrolyte. In the bath, current is transferred in the electrolyte by ionic conduction. The potential applied between anode and cathode is the driving force for transfer of charge across the metal-solution interfaces (2–16 V), e.g. electro deposition of copper from copper sulphate:
CuSO4→ Cu2++ SO2-4
Manufacture:
- Electroplating readily lends itself to unit, batch or continuous production techniques.
- Depositing operations can be performed manually, or by means of semi or fully automated handling equipment. Another method is barrel plating.
- In all plant, except barrel plating of small components, all parts to be plated are individually mounted on jigs or wires. Areas not requiring plate are coated with stopping-off agents.
- Automated, high volume electroplating plant essentially consists of a line of process tanks with a transfer system which enables the jigs to be immersed in each tank for the appropriate length of time, and in the sequence necessary to achieve the required finish. The incorporation of programmable sequential controllers enables the process times to be modified as required for a wide variety of components, and the provision of overhead transporters on tracks provides a flexible manufacturing system.
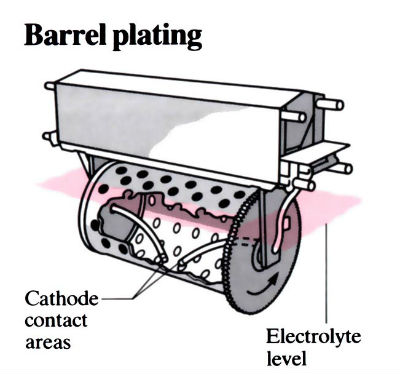
- Barrel plating is widely employed for the bulk processing of small to medium-sized components. Multi-station units can be employed to complete all operations in the one line, including pre- and post-plating operations. Perforated barrels are immersed in the plating solution (sizes range from 200 mm to 1 m in length) and are made out of G76 polypropylene with the anodes placed outside the barrel. The barrels can operate up to 82°C. The barrels are rotated at speeds of 5–15 rev min-1.
Materials:
- Metals which are electroplated include nickel, chromium, tin, copper, silver, zinc, lead and gold.
- Alloys which are electrodeposited include brass (Cu-Zn), bronze (Cu-Sn), lead tin and tin nickel.
- Nickel and chromium coatings predominate, singly or combined, for mainly decorative and corrosion-resistant finishes. Duplex coatings have two layers of nickel and one top layer of chromium. Deposit thicknesses depend on application, but nickel is usually 5–30 μm and the chromium top layer is 5–8 μm. Chromium can be a bright or black finish.
- Additions to bright nickel solutions for nickel plating can be used for levelling, so that a deposit modifies any minor surface irregularities such as scratches or polishing marks, which tend to become filled.
- Thicker layers of chromium by itself can be applied to components to increase the wear resistance of the part. This is known as hard chrome-plating, with deposit thicknesses of 10–500 μm achieving hardnesses of 950–1000 VPN.
- Tin is electrodeposited to 5–12 μm thicknesses for steel cans for non-toxic use with foodstuffs.
- Silver (5–50 μm) is plated onto electrical connectors because of its high thermal and electrical conductivity.
- Zinc (5–30 μm) gives good corrosion protection to steel (as an alternative to galvanising, which is mostly used for large surface areas).
Design:
- Component design for engineering plating should avoid sharp contours wherever possible. Correct control of the plating process, in connection with making allowance for variable “throwing powers” on a contoured shape, can reduce the variations in thicknesses that can occur on electroplating.
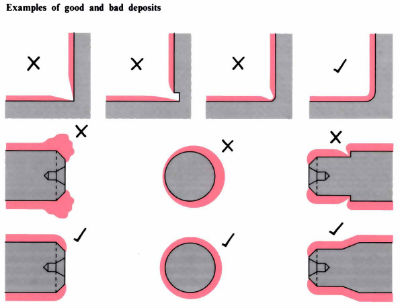
- Wherever possible, consideration should be given to plating to a final size and tolerance. Thicknesses of deposits depend on application. Thin coatings (25–50 μm) tend to be for decorative and corrosion resistance applications, whereas thicker coatings (50–500 μm) tend to be used for wear resistance or metal build-up.
- Deposits > 125 μm require final grinding to obtain final size tolerances.
- Cathodic deposition is accompanied by release of a small amount of hydrogen, which might diffuse into the workpiece. When plating high hardness steel, the parts are heated to 200°C after plating, to prevent H2 embrittlement.
See also: Electroless plating, Anodising, Phosphating, Toyota diffusion (TD) and Chemical vapour deposition (CVD).
This article is a part of Manupedia, a collection of information about some of the processes used to convert materials into useful objects.
Rate and Review
Rate this article
Review this article
Log into OpenLearn to leave reviews and join in the conversation.
Article reviews