5.3.1 Muscles
The rates of muscle contraction and relaxation, and the maximum force generated, are complex enzymatic processes that determine speed of swimming. Ian Johnston of St Andrews University (Johnston, 1989) has compared the maximum tension of muscle fibres isolated from several species of antarctic, temperate-zone and tropical fish (Figure 25).
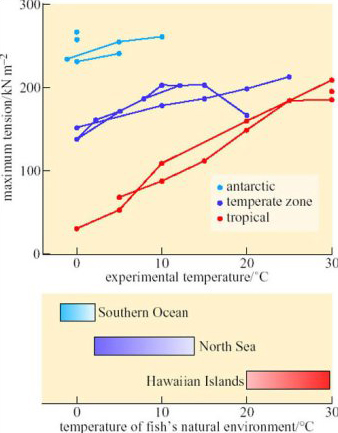
SAQ 39
Do the data on Figure 25 provide evidence for temperature compensation of the contractile mechanism?
Answer
Yes. Between 0 and 10° C, muscles from antarctic fish generate forces five to ten times larger than those measured from muscles of tropical species.
However, these properties of isolated muscle fibres did not match well with studies of individual molecules and whole animals. Temperature compensation could not be demonstrated in the maximum activity of some key enzymes in muscle contraction (e.g. ATPase) and many mitochondrial enzymes (e.g. cytochrome oxidases) studied in vitro. When temperate-zone fish such as goldfish (Carassius auratus), eels (Anguilla anguilla) and carp (Carassius carassius) are acclimated to low temperatures, the proportion of the volume of red muscle fibres occupied by mitochondria increases from 14% at 28° C to 25% at 2° C, indicating that oxidative capacity is maintained by the presence of more mitochondria, rather than by temperature compensation of the enzymes. Antarctic fish also generally have more and/or larger mitochondria but the data are not very clear cut: the proportion of the volume of red muscle fibres occupied by mitochondria ranges from 13–56% in the five antarctic species studied, compared to 4–45% in temperate-zone fish. The muscle fibres of the icefish (Chaenocephalus aceratus, Figure 26a) are more than half mitochondria, leaving little room for the contractile mechanism itself. Those of red-blooded antarctic fish, such as Notothenia gibberifrons (Figure 26b), contain a greater proportion of contractile myofibrils than the icefish, but mitochondria are still more abundant than in temperate-zone fish, particularly towards the edges of the muscle fibres near the blood vessel.

SAQ 40
How could mitochondria arranged as in Figure 26 adapt the muscles to activity at low temperature?
Answer
Diffusion is slower in the cold. Delays in metabolites such as ATP reaching the contractile proteins are minimized if numerous mitochondria are interspersed between the muscle fibres, thereby shortening the mean distance between the mitochondria and the ATP-using enzymes.
Biologists from the University of Maine compared the maximum activities at 1° C of several enzymes in the swimming muscles and the heart of two antarctic fish, Notothenia gibberifrons (similar to Figure 23b) and Trematomus newnesi (similar to Figure 23c), with those of two species of similar size and habits caught in the western Atlantic Ocean off the coast of Delaware, USA. They found that the activities of enzymes involved in lipid catabolism and aerobic respiration, such as carnitine palmitoyltransferase and 3-hydroxyacyl CoA dehydrogenase, were 1.3–27.0 times higher in the red muscles of the antarctic species than in those of the temperate-zone species and the Q10 values were less than 2. However, the activities of phosphofructokinase, pyruvate kinase and lactate dehydrogenase that are essential to anaerobic utilization of carbohydrates were either not significantly different or were lower in the polar species. Thus these antarctic fish seem to be equipped to use lipid fuels more efficiently than carbohydrate fuels.
Maximum swimming speed during brief ‘bursts’ of activity (e.g. when escaping from a predator) has been measured accurately in only two species of antarctic fish, and was found to be at the lower end of the range found in temperate-zone fish of similar size and body shape. Clearly, the situation is complicated and further research on a greater range of species is necessary to understand adaptation to low temperatures. Nonetheless, even such limited information enables us to identify some principles of adaptation to polar conditions in ectotherms.
SAQ 41
With which of the two mechanisms for maintaining ionic balance at low temperature are these data most consistent?
Answer
Taken together, the observations suggest that antarctic fish living in surface waters achieve temperature compensation by increased activity of the ionic pump.
SAQ 42
Why would this mechanism be better for antarctic fish living in surface waters?
Answer
Slow movement is unlikely to be adaptive where fast-swimming, endothermic predators such as penguins and seals are about. There are relatively few surface-swimming fish in antarctic waters. Most fish live on or near the bottom, or in deep waters, out of reach of most air-breathing predators.
The silver fish (Pleuragramma antarcticum; Figure 23a) lives in the surface layers of coastal waters and eats pelagic invertebrates. Around McMurdo Sound in Antarctica, silver fish are known to be an important food for penguins, skuas and Weddell seals (Leptonychotes weddellii). Although its high density of mitochondria must increase its BMR and the energy cost of swimming, such adaptations to quick responses and fast escape are probably essential to avoiding predation. In contrast, most deep-sea fishes that have been investigated (and only a few species have been kept alive in surface laboratories for long enough to be studied) have lower BMR than expected and probably compensate for low temperature by reducing membrane permeability. Mammalian and avian predators are absent in the deep sea, and food (and probably also oxygen) is scarce, so the alternative mechanism for maintaining the potential gradient across the cell membranes is more efficient.
Another peculiar and consistent feature of nototheniid fish is that most species, including P. antarcticum (Figure 23a), Dissostichus mawsoni (Figure 23d) and species of Notothenia and Trematomus, have numerous sacs of lipid within and around their swimming muscles and between the muscles and the skin. Several functions have been suggested: energy stores, buoyancy (which could be important in fish that lack swim bladders) and more recently, oxygen diffusion. The solubility of oxygen in lipid is four times higher than in aqueous cytoplasm, and, particularly in fish in which blood pigments are reduced or absent, lipids closely associated with muscles may facilitate oxygenation of the tissues.
You may have noticed that in Figure 25 the contraction of muscles from tropical and temperate-zone fish were measured over a temperature range from 0° C to 25–30° C, but there are no data for the muscles of antarctic fish above 10° C. Many polar fish tolerate only a very narrow range of temperatures and die within minutes if warmed more than a few degrees above the temperature of the water in which they normally live. Exactly why they die is not clear, but a likely cause is widely different Q10 values of enzymes in critical metabolic pathways, such as ion pumps or mitochondria: a small change of temperature puts the whole pathway ‘out of kilter’, causing metabolic intermediates to accumulate to toxic concentrations. In this respect, polar fish resemble non-hibernating homeotherms such as humans and rats: their metabolism is seriously, often irreversibly, disrupted by small departures from their normal body temperature. This property, of course, makes it much more difficult to transport such fish alive and to keep them in captivity for long enough to study habits such as breeding, growth and dietary preferences.
