3.9 Summary
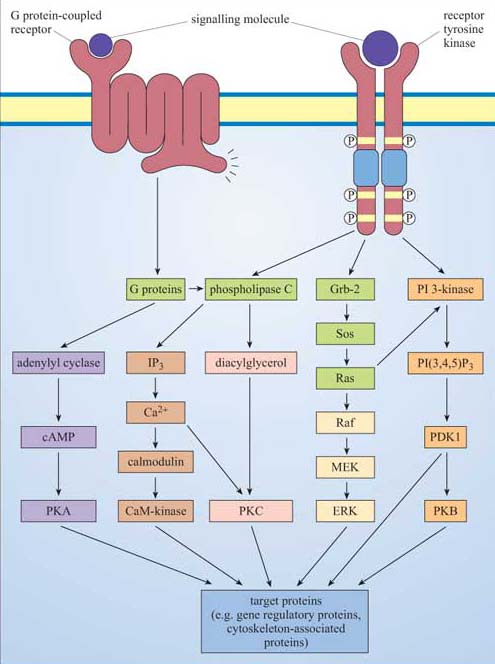
Heterotrimeric G proteins are tethered to the internal surface of the plasma membrane, and are activated by conformational change within 7TM receptors. There are many different α subunits (and a few βγ subunits), which interact with different receptors and different effectors. The major targets of G proteins include ion channels, adenylyl cyclase (activated by Gαs and inhibited by Gαi) and PLC-β (activated by Gαq).
Phosphatidylinositol (PI) is the precursor of a family of small lipid second messengers. The inositol ring can be further phosphorylated at positions 3, 4, and 5 by lipid kinases. PI 3-kinase specializes in phosphorylating the hydroxyl group at the 3 position, thus generating the active signalling molecules PI(3,4)P2 and PI(3,4,5)P3. The phosphorylated 3 position is recognized as a docking site by PH domain-containing proteins, thus providing a mechanism for signalling proteins to be recruited to the membrane.
Phospholipase C enzymes (especially PLC-β, activated by G proteins, and RTK-activated PLC-γ) cleave PI(4,5)P2 to generate diacylglycerol (DAG) and inositol 1,4,5-triphosphate (IP3). DAG remains embedded in the membrane, where it activates protein kinase C (PKC). IP3 diffuses through the cytosol, and opens IP3-gated calcium channels, releasing stored calcium into the cytosol.
The Ca2+ ion is an important second messenger, which enters the cytosol from the extracellular space through specific channels on the plasma membrane, or is rapidly released from stores into the cytoplasm. Calcium channels include IP3-gated calcium channels, voltage-dependent calcium channels, or ryanodine receptors in skeletal muscle cells. It activates numerous Ca2+ -dependent proteins, including PKC, but many of its effects are mediated via calmodulin, which has four allosteric Ca2+ binding sites. Ca2+ /calmodulin then binds to and regulates target proteins, especially Ca2+ /calmodulin-dependent protein kinases (CaM kinases).
Cyclic AMP (cAMP) is another important second messenger, synthesized by adenylyl cyclase (which is activated or inhibited by different G protein subtypes). It can open cAMP-gated ion channels, but it mediates many of its effects through cAMP-dependent protein kinase A (PKA), whose roles include regulating glycogen metabolism, and phosphorylation of a transcription factor that binds to the cAMP response element (CRE).
Cyclic GMP is synthesized by guanylyl cyclase. Its targets include cGMP-gated ion channels and a cGMP-dependent kinase (PKG).
Ras is the archetypal monomeric, or small, G protein. Ras classically operates downstream of growth factor receptors: Grb-2, an SH2/SH3-containing adaptor protein, binds to phosphotyrosines on the activated RTK, and recruits Sos to the membrane environment; Sos promotes GTP binding by Ras. Activated Ras has more than one target, including PI 3-kinase, but its most important downstream pathway is the MAP kinase pathway. Activated Ras recruits Raf to the membrane, where it is activated and then phosphorylates MEK, which then phosphorylates ERK, a MAP kinase. These have multiple cytoplasmic and transcription factor targets involved in cell growth and division or differentiation.
Protein kinase families involved at various points in signalling pathways include receptor tyrosine kinases (for example, the EGF receptor), non-receptor tyrosine kinases (such as Src and JAK), serine–threonine kinases such as PKC, PKA, MAP kinases and the TGF receptor, and rare dual-specificity kinases such as MEK.
Protein phosphatases dephosphorylate proteins, and are grouped according to their targets, as are protein kinases. They include protein tyrosine phosphatases, serine–threonine phosphatases, and a few dual-specificity phosphatases.
The duration of ERK activity determines activation of different transcription factors (SRF/TCF for immediate early genes; AP-1 for other target genes).
The major signalling molecules we have discussed are brought together into five basic pathways in Figure 46.