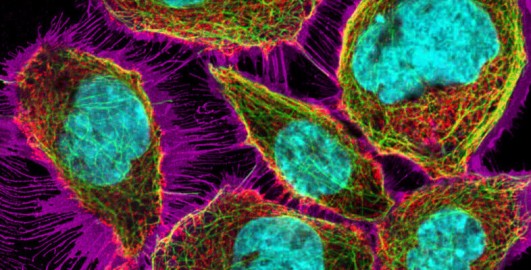
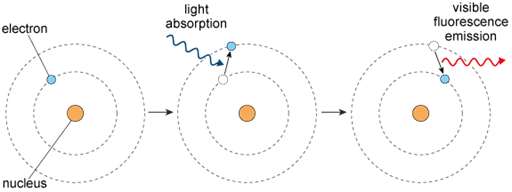
3.1 Fluorescence is activated by light
What makes fluorescent molecules like GFP ‘glow’ the way they do? To understand this, it is necessary to look at the molecular level and specifically at the arrangements of atoms. Atoms contain a central nucleus around which electrons move in electron shells (or orbitals). The energy state of electrons increases from the innermost to the outer shells.
Fluorescent molecules, also called fluorophores, generally contain rings of carbon atoms (called aromatic rings). These molecules can absorb incoming energy (usually in the form of light). As a result of the absorption of energy, electrons within the molecules change from a resting condition called the ‘ground state’ to an ‘excited state’ on a shell further away from the nucleus for a very brief period of time. Eventually, the excited electrons return to their ground state, and as they do this, they emit some of the energy they had originally absorbed as photons of light (Figure 9).