4.2 Using dyes (or probes) to stain structures
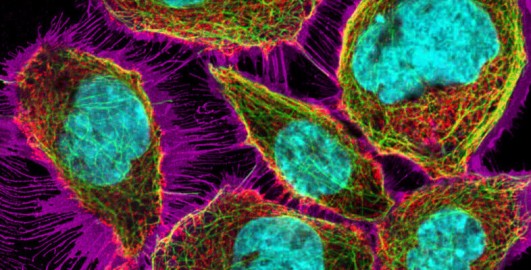
You learned about fluorescent molecules (also known as fluorophores) earlier in this course, and that it is possible to visualise several structures inside cells at the same time using fluorophores that emit different colours of light (Figure 11).
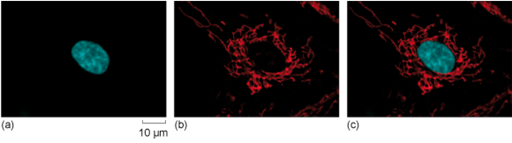
If you want to visualise red fluorescence, like the one in mitochondria shown in Figure 11, why would you use green light to excite the fluorophore?
Answer
Referring back to the spectrum of visible light shown in Figure 10, and the principle of fluorescence, the excitation wavelength must be shorter than the emission wavelength. Green light has a shorter wavelength and more energy, so it can be used to cause emission of red light.
Staining of structures can be done with, for example, fluorescent dyes, stains, probes, or labels. These terms are often used interchangeably in science literature. Fluorescent indicators change their properties, for example their brightness, depending on their environment. For example, they get brighter when the concentration of a certain ion changes.
The concentration of which ion can be measured by a fluorescent indicator that can show the change of the intracellular pH value?
Answer
The concentration of ions (protons). An increase in the proton concentration causes a drop in the pH (it becomes more acidic). A decrease in the proton concentration causes an increase of the pH (it becomes more alkaline).
Many fluorescent dyes and fluorescent indicators are designed to be membrane-permeable and can be taken up by living cells by simply immersing a tissue section, or cells grown on a glass coverslip, in a solution containing the dye or indicator, and then rinsing off the excess.