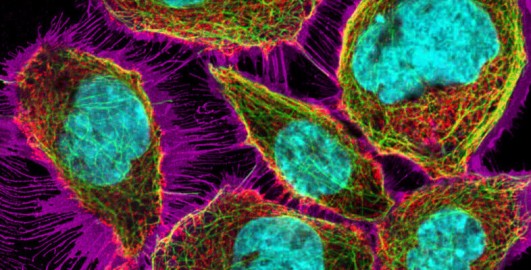
4.3 Immunolabelling
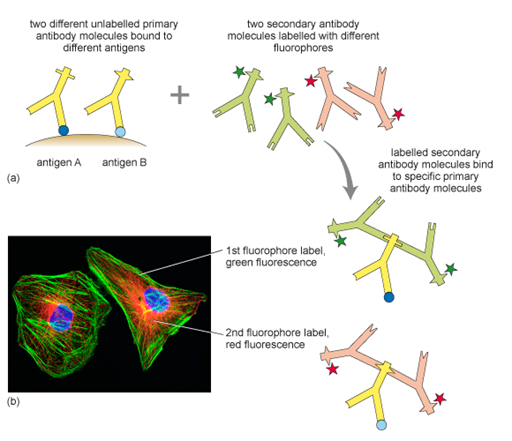
Immunolabelling is a technique used to detect specific proteins or structures in cells or tissues by using antibodies — molecules that naturally recognise and bind to particular targets. A so-called primary antibody (yellow Y-shapes in Figure 12a) binds to a target, for example the protein actin. Primary antibodies are raised in different animals. The so-called primary antibody is either directly attached to a fluorescent dye or detected by a second fluorescently labelled antibody (green and red Y-shapes in Figure 12a). When viewed under a fluorescence microscope, the dye lights up, allowing researchers to see where the protein is located in the sample. A common variation of this technique is double immunolabelling, where two different proteins are labelled at the same time using antibodies tagged with two distinct fluorescent dyes, or by using two primary antibodies that were raised in different animal species. These primary antibodies are recognised specifically by different secondary antibodies, labelled with different fluorophores.
Double immunolabelling allows scientists to observe the spatial relationship or co-localisation of different proteins within the same cell or tissue (Figure 12). Immunolabelling can only be performed in fixed and permeabilised cells, because the large antibody molecules would otherwise not be able to enter the cells. By using labels other than fluorophores, immunolabelling can also be visualised with light and electron microscopy.
Double immunolabelling to detect two proteins, A and B, would not work if the primary antibodies for A and B were both mouse antibodies. Can you suggest why?
Answer
Each protein would bind a mouse primary antibody. The anti-mouse secondary antibody subsequently applied to detect the primary antibodies would then bind to both primaries, and so they would both be labelled with the same detection molecule (e.g. the same colour of fluorescence) and it would not be possible to see the separate locations of proteins A and B.