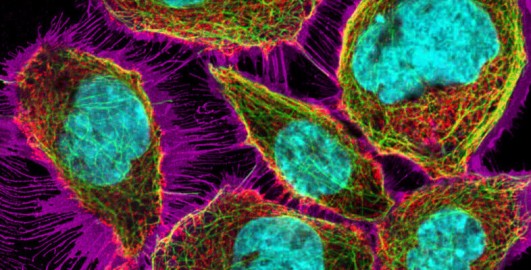
4.5 Similarities and differences between the three types of microscopy
After learning about three types of microscopy that are often used to study cells, and about commonly used different ways to make structures in cells fluorescent, you are now encouraged to test your learning and understanding by filling in the following table and answering the quiz questions.
Using your knowledge from this course, fill in Table 2. To give you an idea how much detail to include, the first column has been filled in. You will find a filled-in version in the answer underneath.
| Feature | Light microscopy | Fluorescence microscopy | Electron microscopy |
|---|---|---|---|
Source of illumination |
Visible white light |
||
Resolution |
~ 200 nm |
||
Magnification (maximum) |
Up to ~1,000× |
||
Staining required? |
Often needed (e.g. dyes) |
||
Live imaging? |
Yes |
||
Contrast? |
Low (without stains) |
||
Is specific labelling possible? |
Limited (general stains) |
||
Type of image produced |
Brightfield image (natural contrast or stained) |
||
Sample preparation |
Simple and fast |
||
Main applications |
Basic cell structure, tissues |
Answer
Here is a filled-in version of the table with information from this course.
| Feature | Light microscopy | Fluorescence microscopy | Electron microscopy |
|---|---|---|---|
Source of illumination |
Visible white light |
Specific wavelengths of light (often UV or laser) |
Beam of electrons |
Resolution |
~ 200 nm |
~ 200 nm |
~0.1–1 nm (much higher due to electron wavelength) |
Magnification |
Up to ~1,000× |
Up to ~1,000× |
Up to ~1,000,000 for TEM Up to ~2,000,000 for SEM |
Staining required? |
Often needed (e.g. dyes) |
Yes, fluorescent dyes or proteins |
Yes, heavy metals |
Live imaging? |
Yes |
Yes |
No (requires vacuum and fixed/dehydrated samples) |
Contrast? |
Low (without stains) |
High (fluorescent signal on dark background) |
Very high due to electron scattering |
Is specific labelling possible? |
Limited (general stains) |
Yes, highly specific using antibodies or tagged proteins |
Limited; specific structures identified by morphology |
Type of image produced |
Brightfield image (natural contrast or stained) |
Fluorescent image on dark background |
Black and white |
Sample preparation |
Simple and fast |
Moderate (labelling, fixing if needed) |
Complex, time-consuming |
Main applications |
Basic cell structure, tissues |
Protein localisation, live-cell imaging, molecular tracking |
Ultrastructure, organelles, viruses, nanostructures |
You are part of a research team investigating the localisation and function of a specific protein in nerve cells. In your studies, you want to study different aspects of this protein. Unless specifically mentioned, the protein has not been genetically modified to carry a fluorescent marker. You have access to three types of microscopes: a standard light microscope, a fluorescence microscope, and an electron microscope. Questions 1-4 explain experiments your research team wants to perform. Based on the features of each microscopy technique (illumination, resolution, sample preparation, and ability to image live cells), which microscope would you choose for each of the experiments and why?
Question 1
Your goal is to identify if this protein is found in ribosomes, which have a diameter of around 25 nm.
Answer
To identify if the protein is localised in ribosomes, using the electron microscope is the most suitable option. Here’s why:
Resolution:
The electron microscope is the only one of the three types that has a high enough resolution to visualise ribosomes. It has a resolution of 0.1 – 1 nm.
Specific labelling:
You would use immunolabelling to visualise the protein in the cell, and you would identify ribosomes by their characteristic appearance.
Live-cell imaging:
To identify the localisation, you don’t need to work with living cells. Fixing the samples during the preparation for electron microscopy ensures that structures and proteins are found in the place where they were present in the living cell.
Question 2
Your goal is to identify if this protein is present in lysosomes, which have a diameter of 0.5 – 1 µM. Your research team commonly uses a fluorescent dye that you know specifically labels lysosomes.
Answer
To identify if a specific protein is present in lysosomes, which can be identified with a fluorescent dye, using the fluorescence microscope combined with immunolabelling is the most suitable option. Here’s why:
Specific labelling:
The protein is not tagged with a fluorescent marker, which means you need to visualise it using immunolabelling. Lysosomes can be specifically labelled with the fluorescent dye, so you want to visualise your immunolabelling with fluorescence microscopy. Light microscopy cannot visualise your protein with such high specificity.
Live-cell imaging:
For the purpose of this experiment, you cannot use live cell imaging because the process of immunolabelling needs the cells to be fixed and permeabilised.
Resolution:
The resolution of fluorescence microscopy is ~200 nm, which is sufficient for visualising lysosomes.
Question 3
Your goal is to track this protein in real time to understand how its behaviour changes in response to different stimuli. For this experiment, the protein has been genetically tagged with a fluorescent marker.
Answer
To track a specific protein in real time within living nerve cells, using the fluorescence microscope is the most suitable option. Here’s why:
Specific labelling:
The protein has been tagged with a fluorescent marker, which means fluorescence microscopy is required to excite and detect that specific signal. Neither light microscopy nor electron microscopy can visualise tagged proteins with such specificity.
Live-cell imaging:
Fluorescence microscopy allows imaging of living cells. Electron microscopy cannot be used for live samples, as it requires the sample to be fixed, dehydrated, and placed in a vacuum. Light microscopy can image live cells but cannot track specific proteins unless general staining is sufficient, which isn’t in this case.
Real-time observation:
Fluorescence microscopy can capture dynamic processes over time, making it ideal for tracking the movement of proteins or organelles within cells.
Resolution:
While not as high as electron microscopy, the resolution (~200 nm) is sufficient for visualising protein localisation and movement within the broader cellular context, especially with the help of time-lapse imaging or even super-resolution techniques if needed.
Question 4
You have already confirmed that cells from individuals with a certain disease express a higher level of the protein you are studying. Now you want to know if the overall cell structure is changed in cells from individuals with the disease. You don’t need to visualise the protein itself.
Answer
Changes in cellular structure, without the need to identify specific proteins, is best done with a light microscope. Here’s why:
Specific labelling:
Light microscopes are not good to visualise specific labelling due to the lower contrast of the samples. However, specific labelling is not necessary in this study, and the sample preparation for light microscopy is often quicker and cheaper than that for the other techniques. That’s why light microscopy is often used in pathology labs to study changes in cellular structure in disease development.
Live-cell imaging:
There is no need for live-cell imaging in this study.
Resolution:
Studying changes in the overall cellular structure does not need a high resolution, so light microscopy is suitable.